Neurite Outgrowth诱导Drimane型倍半萜类化合物从紫外多孢菌MUCL 56355的培养物中分离。
IF 3.3
2区 生物学
Q2 CHEMISTRY, MEDICINAL
引用次数: 1
摘要
Abundaporin A(1)和7个以前未描述的drimane倍半萜,命名为Abundaporens B-H(2-8),是从肯尼亚采集的一个多孔菌Abundaporus violaceus MUCL 56355(多孔菌科)中分离出来的。基于详尽的1D和2D NMR光谱测量并得到HRESIMS数据的支持,阐明了分离的化合物的化学结构。分离的化合物的绝对构型通过对1-4使用Mosher方法和对4和5-8使用TDDFT-ECD计算来确定。在抗微生物或细胞毒性测定中,没有一种分离的化合物表现出显著的活性。值得注意的是,所有测试的化合物都表现出神经营养作用,当用5ng/mL NGF治疗时,1和6显著增加了轴突的生长。本文章由计算机程序翻译,如有差异,请以英文原文为准。
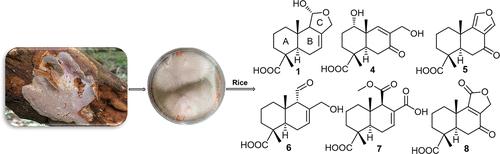
Neurite Outgrowth-Inducing Drimane-Type Sesquiterpenoids Isolated from Cultures of the Polypore Abundisporus violaceus MUCL 56355
Abundisporin A (1), together with seven previously undescribed drimane sesquiterpenes named abundisporins B–H (2–8), were isolated from a polypore, Abundisporus violaceus MUCL 56355 (Polyporaceae), collected in Kenya. Chemical structures of the isolated compounds were elucidated based on exhaustive 1D and 2D NMR spectroscopic measurements and supported by HRESIMS data. The absolute configurations of the isolated compounds were determined by using Mosher’s method for 1–4 and TDDFT-ECD calculations for 4 and 5–8. None of the isolated compounds exhibited significant activities in either antimicrobial or cytotoxicity assays. Notably, all of the tested compounds demonstrated neurotrophic effects, with 1 and 6 significantly increasing outgrowth of neurites when treated with 5 ng/mL NGF.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
9.10
自引率
5.90%
发文量
294
审稿时长
2.3 months
期刊介绍:
The Journal of Natural Products invites and publishes papers that make substantial and scholarly contributions to the area of natural products research. Contributions may relate to the chemistry and/or biochemistry of naturally occurring compounds or the biology of living systems from which they are obtained.
Specifically, there may be articles that describe secondary metabolites of microorganisms, including antibiotics and mycotoxins; physiologically active compounds from terrestrial and marine plants and animals; biochemical studies, including biosynthesis and microbiological transformations; fermentation and plant tissue culture; the isolation, structure elucidation, and chemical synthesis of novel compounds from nature; and the pharmacology of compounds of natural origin.
When new compounds are reported, manuscripts describing their biological activity are much preferred.
Specifically, there may be articles that describe secondary metabolites of microorganisms, including antibiotics and mycotoxins; physiologically active compounds from terrestrial and marine plants and animals; biochemical studies, including biosynthesis and microbiological transformations; fermentation and plant tissue culture; the isolation, structure elucidation, and chemical synthesis of novel compounds from nature; and the pharmacology of compounds of natural origin.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


