设计和合成新型吲哚衍生 N-甲基氨基甲酰基胍基几丁质酶抑制剂,显著提高杀虫活性
IF 5.7
1区 农林科学
Q1 AGRICULTURE, MULTIDISCIPLINARY
引用次数: 0
摘要
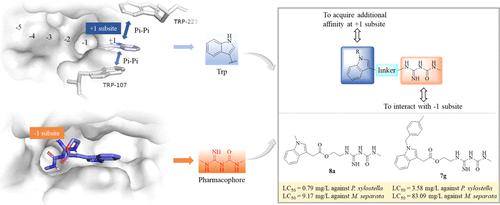
几丁质酶在昆虫蜕皮过程中发挥着重要作用,是开发绿色杀虫剂的潜在靶标。根据OfChtI、OfChtII和OfChi-h中+1/+2位点的色氨酸残基处于错配平行位置的特点,提出了在几丁质酶抑制剂中引入吲哚支架的策略,并成功合成了含有N-甲基氨基甲酰基胍基和吲哚支架的多靶点几丁质酶抑制剂。抑制活性表明,化合物 8u 对 OfChtI、OfChtII 和 OfChi-h 具有显著的抑制活性,IC50 值分别为 0.7、0.79 和 0.58 μM,Ki 值分别为 0.05 ± 0.005、0.065 ± 0.004 和 0.025 ± 0.006 μM。体内杀虫活性表明,化合物 8a 和 8g 对木虱(Plutella xylostella)和绣线菊(Mythimna separata)具有很好的杀虫活性,对木虱的半致死浓度分别为 0.79 和 9.17 mg/L,对绣线菊的半致死浓度分别为 3.58 和 83.09 mg/L,是迄今为止发现的具有体内杀虫活性的最强几丁质酶抑制剂。抑制机理和结合自由能结果表明,N-甲基氨基甲酰基胍基与-1催化位点结合,而通过π-π堆积和吲哚支架与色氨酸的疏水相互作用获得的额外相互作用增加了靶标与几丁质酶的结合亲和力。这项工作为几丁质酶抑制剂的开发提供了一个新方向,化合物 8a 和 8g 有可能成为农药开发的候选化合物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Design and Synthesis of Novel Indole-Derived N-Methylcarbamoylguanidinyl Chitinase Inhibitors with Significantly Improved Insecticidal Activity
Chitinases play an important role in the molting process of insects and are potential targets for the development of green insecticides. Based on the feature that the +1/+2 sites in OfChtI, OfChtII, and OfChi-h have tryptophan residues in mismatch-parallel position, a strategy to introduce indole scaffold into chitinase inhibitors was proposed, and multitarget chitinase inhibitors containing N-methylcarbamoylguanidinyl and indole scaffold were successfully synthesized. The inhibitory activity showed that compound 8u exhibited significant inhibitory activity against OfChtI, OfChtII, and OfChi-h, with IC50 values of 0.7, 0.79, and 0.58 μM, and Ki values of 0.05 ± 0.005, 0.065 ± 0.004, and 0.025 ± 0.006 μM, respectively. In vivo insecticidal activity showed that compounds 8a and 8g exhibited excellent insecticidal activity against Plutella xylostella and Mythimna separata, with LC50 values of 0.79 and 9.17 mg/L against P. xylostella, respectively, and 3.58 and 83.09 mg/L against M. separata, respectively, making them the most potent chitinase inhibitors with in vivo insecticidal activity discovered to date. The inhibition mechanism and binding free energy results suggested that N-methylcarbamoylguanidinyl binds to the −1 catalytic site, while additional interactions acquired by π–π stacking and hydrophobic interactions of the indole scaffold with tryptophan increase the binding affinity of the targets to chitinases. This work provides a new direction for the development of chitinase inhibitors with compounds 8a and 8g potentially serving as promising candidates for pesticide development.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
9.90
自引率
8.20%
发文量
1375
审稿时长
2.3 months
期刊介绍:
The Journal of Agricultural and Food Chemistry publishes high-quality, cutting edge original research representing complete studies and research advances dealing with the chemistry and biochemistry of agriculture and food. The Journal also encourages papers with chemistry and/or biochemistry as a major component combined with biological/sensory/nutritional/toxicological evaluation related to agriculture and/or food.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


