作为杀虫剂的吡啶衍生物:第 6 部分。3-(取代)甲硫基-5,6,7,8-四氢异喹啉-4-甲腈的设计、合成、分子对接和对蚜虫(Glover,1887 年)的杀虫活性
IF 5.7
1区 农林科学
Q1 AGRICULTURE, MULTIDISCIPLINARY
引用次数: 0
摘要
6-二甲基-6-羟基-8-(4-吡啶基、3-吡啶基、苯基、4-甲氧基苯基或 4-氯苯基)-5,6,7,8-四氢异喹啉-3(2H)-硫酮 2a-e 的高产率和高纯度设计与合成。因此,在温和的碱性条件下,化合物 2a-e 与一些氯试剂,即 N-芳基-2-氯乙酰胺 3a-f 和 N-(萘-2-基)-2-氯乙酰胺(3g)反应,分别得到前两个系列的目标化合物 3-(N-芳基)氨基甲酰甲硫基-5,6,7,8-四氢异喹啉-4-甲腈 4a-l 和 5a-e。在相同条件下,化合物 2d、e 与氯乙酸乙酯反应,得到了另一个系列的 3-乙氧羰基甲硫基-5,6,7,8-四氢异喹啉-4-甲腈 6d、e。筛选了所有合成的 5,6,7,8-四氢异喹啉类化合物对蚜虫若虫和成虫的杀虫活性。结果表明,一些测试化合物具有良好的杀虫活性。此外,还对一些代表性化合物的结构-活性关系以及分子对接进行了评估。本文章由计算机程序翻译,如有差异,请以英文原文为准。
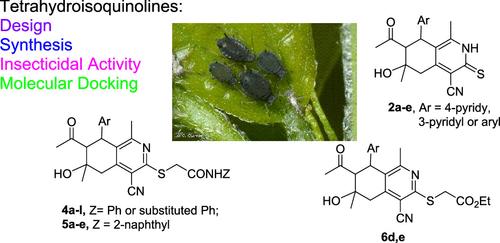
Pyridine Derivatives as Insecticides: Part 6. Design, Synthesis, Molecular Docking, and Insecticidal Activity of 3-(Substituted)methylthio-5,6,7,8-tetrahydroisoquinoline-4-carbonitriles Toward Aphis gossypii (Glover, 1887)
Three new series of 3-(substituted)methylthio-4-cyano-5,6,7,8-tetrahydroisoquinolines were designed and synthesized starting from readily available materials, 7-acetyl-4-cyano-1,6-dimethyl-6-hydroxy-8-(4-pyridyl, 3-pyridyl, phenyl, 4-methoxyphenyl, or 4-chlorophenyl)-5,6,7,8-tetrahydrosoquinoline-3(2H)-thiones 2a–e in high yields and very pure states. Thus, compounds 2a–e were reacted with some chloro reagents, namely, N-aryl-2-chloroacetamides 3a–f and N-(naphthalen-2-yl)-2-chloroacetamide (3g) under mild basic conditions to give the first two series of the target compounds, 3-(N-aryl)carbamoylmethylthio-5,6,7,8-tetrahydroisoquinoline-4-carbonitriles 4a–l and 5a–e, respectively. Reaction of compounds 2d,e with ethyl chloroacetate under the same conditions gave the other series, 3-ethoxycarbonyl-methylthio-5,6,7,8-tetrahydroisoquinoline-4-carbonitriles 6d,e. Structural formulas of all of the new compounds were elucidated and confirmed by elemental and spectral analyses. The insecticidal activity of all synthesized 5,6,7,8-tetrahydrosoquinolines toward the nymphs and adults of Aphis gossypii were screened. The results revealed the promising insecticidal activity of some tested compounds. Moreover, the structure–activity relationships as well as molecular docking of some representative compounds were evaluated.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
9.90
自引率
8.20%
发文量
1375
审稿时长
2.3 months
期刊介绍:
The Journal of Agricultural and Food Chemistry publishes high-quality, cutting edge original research representing complete studies and research advances dealing with the chemistry and biochemistry of agriculture and food. The Journal also encourages papers with chemistry and/or biochemistry as a major component combined with biological/sensory/nutritional/toxicological evaluation related to agriculture and/or food.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


