从一种海洋衍生真菌 Arthrinium arundinis 菌株中提取的具有抗炎和神经保护作用的 α-Pyrones 及其异源表达
IF 3.6
2区 生物学
Q2 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
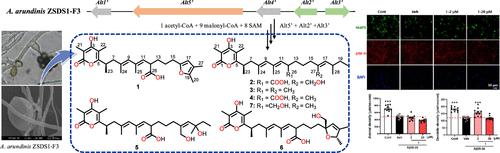
真菌线性多酮(如带有 6-烯基链的 α-吡喃酮)是生物活性化合物的丰富来源。研究人员从一种源自海洋的 Arthrinium arundinis 真菌菌株中分离出了两种新的(1 和 2)和四种已知的(3-6)6-烯基吡喃酮多酮类化合物。通过大量光谱分析确定了它们的结构。通过在黑曲霉 A1145 ΔSTΔEM 中的异源表达验证了交替吡喃酮的生物合成基因簇(alt),发现细胞色素 P450 单加氧酶 Alt2′ 可以将甲基 26-CH3 转化为羧基,从 3 生成 4。另一种细胞色素 P450 单加氧酶 Alt3′ 可催化连续的羟化、环氧化和氧化步骤,从 4 生成 1、2、5 和 6。交替吡喃酮 G(1)不仅能抑制脂多糖(LPS)刺激的 BV2 小胶质细胞的 M1 极化,还能刺激 Aβ 处理后的树突再生和神经元存活,这表明交替吡喃酮 G 可被用作阿尔茨海默氏症药物研发的重要支架。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Anti-inflammatory and Neuroprotective α-Pyrones from a Marine-Derived Strain of the Fungus Arthrinium arundinis and Their Heterologous Expression
Fungal linear polyketides, such as α-pyrones with a 6-alkenyl chain, have been a rich source of biologically active compounds. Two new (1 and 2) and four known (3–6) 6-alkenylpyrone polyketides were isolated from a marine-derived strain of the fungus Arthrinium arundinis. Their structures were determined based on extensive spectroscopic analysis. The biosynthetic gene cluster (alt) for alternapyrones was identified from A. arundinis ZSDS-F3 and validated by heterologous expression in Aspergillus nidulans A1145 ΔSTΔEM, which revealed that the cytochrome P450 monooxygenase Alt2′ could convert the methyl group 26-CH3 to a carboxyl group to produce 4 from 3. Another cytochrome P450 monooxygenase, Alt3′, catalyzed successive hydroxylation, epoxidation, and oxidation steps to produce 1, 2, 5, and 6 from 4. Alternapyrone G (1) not only suppressed M1 polarization in lipopolysaccharide (LPS)-stimulated BV2 microglia but also stimulated dendrite regeneration and neuronal survival after Aβ treatment, suggesting alternapyrone G may be utilized as a privileged scaffold for Alzheimer’s disease drug discovery.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
9.10
自引率
5.90%
发文量
294
审稿时长
2.3 months
期刊介绍:
The Journal of Natural Products invites and publishes papers that make substantial and scholarly contributions to the area of natural products research. Contributions may relate to the chemistry and/or biochemistry of naturally occurring compounds or the biology of living systems from which they are obtained.
Specifically, there may be articles that describe secondary metabolites of microorganisms, including antibiotics and mycotoxins; physiologically active compounds from terrestrial and marine plants and animals; biochemical studies, including biosynthesis and microbiological transformations; fermentation and plant tissue culture; the isolation, structure elucidation, and chemical synthesis of novel compounds from nature; and the pharmacology of compounds of natural origin.
When new compounds are reported, manuscripts describing their biological activity are much preferred.
Specifically, there may be articles that describe secondary metabolites of microorganisms, including antibiotics and mycotoxins; physiologically active compounds from terrestrial and marine plants and animals; biochemical studies, including biosynthesis and microbiological transformations; fermentation and plant tissue culture; the isolation, structure elucidation, and chemical synthesis of novel compounds from nature; and the pharmacology of compounds of natural origin.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


