Rational Modification of a Cross-Linker for Improved Flexible Protein Structure Modeling
IF 6.7
1区 化学
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
Abstract
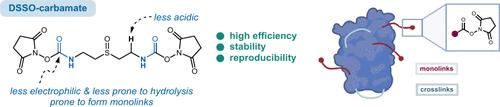
Chemical cross-linking/mass spectrometry (XL-MS) has emerged as a complementary tool for mapping interaction sites within protein networks as well as gaining moderate-resolution native structural insight with minimal interference. XL-MS technology mostly relies on chemoselective reactions (cross-linking) between protein residues and a linker. DSSO represents a versatile cross-linker for protein structure investigation and in-cell XL-MS. However, our assessment of its shelf life and batch purity revealed decomposition of DSSO in anhydrous solution via a retro-Michael reaction, which may reduce the active ingredient down to below 90%. To mitigate the occurrence of this degradative mechanism, we report the rational design and synthesis of DSSO-carbamate, which contains an inserted nitrogen atom in the DSSO backbone structure. This modification to DSSO yielded remarkably favorable stability against such decomposition, which translated to higher cross-link and monolink recovery when performing XL-MS on monomeric flexible proteins. Recently, XL-MS has been leveraged against AlphaFold2 and other protein structure prediction algorithms for improved prediction of flexible monomeric multiconformational proteins. To this end, we demonstrate that our novel cross-linker, termed DSSO-carbamate, generated more accurate protein structure predictions when combined with AlphaFold2, on account of its increased recovery of cross-links and monolinks, compared to DSSO. As such, DSSO-carbamate represents a useful addition to the XL-MS community, particularly for protein structure prediction.
合理修饰交联剂以改进柔性蛋白质结构建模
化学交联/质谱(XL-MS)已经成为一种辅助工具,用于绘制蛋白质网络中的相互作用位点,并以最小的干扰获得中等分辨率的天然结构洞察。xml - ms技术主要依赖于蛋白质残基和连接体之间的化学选择反应(交联)。DSSO是一种多功能交联剂,用于蛋白质结构研究和细胞内xml - ms。然而,我们对其保质期和批次纯度的评估显示,DSSO在无水溶液中通过反向迈克尔反应分解,这可能会将有效成分降低到90%以下。为了减轻这种降解机制的发生,我们报道了在DSSO主链结构中插入氮原子的DSSO-氨基甲酸酯的合理设计和合成。这种对DSSO的修饰对这种分解产生了非常有利的稳定性,当对单体柔性蛋白进行xml - ms时,它转化为更高的交联和单联恢复。最近,xml - ms已经与AlphaFold2和其他蛋白质结构预测算法相结合,以改进对柔性单体多构象蛋白的预测。为此,我们证明了我们的新型交联剂,称为DSSO-氨基甲酸酯,与AlphaFold2结合时产生更准确的蛋白质结构预测,因为与DSSO相比,其交联和单链的恢复增加。因此,dsso -氨基甲酸酯代表了xml - ms社区的一个有用的补充,特别是在蛋白质结构预测方面。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Analytical Chemistry
化学-分析化学
CiteScore
12.10
自引率
12.20%
发文量
1949
审稿时长
1.4 months
期刊介绍:
Analytical Chemistry, a peer-reviewed research journal, focuses on disseminating new and original knowledge across all branches of analytical chemistry. Fundamental articles may explore general principles of chemical measurement science and need not directly address existing or potential analytical methodology. They can be entirely theoretical or report experimental results. Contributions may cover various phases of analytical operations, including sampling, bioanalysis, electrochemistry, mass spectrometry, microscale and nanoscale systems, environmental analysis, separations, spectroscopy, chemical reactions and selectivity, instrumentation, imaging, surface analysis, and data processing. Papers discussing known analytical methods should present a significant, original application of the method, a notable improvement, or results on an important analyte.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


