Efficient Expression and Activity Optimization of Manganese Peroxidase for the Simultaneous Degradation of Aflatoxins AFB1, AFB2, AFG1, and AFG2
IF 5.7
1区 农林科学
Q1 AGRICULTURE, MULTIDISCIPLINARY
引用次数: 0
Abstract
Aflatoxins (AFs), notorious mycotoxins that pose significant risks to human and animal health, make biodegradation extremely crucial as they offer a promising approach to managing and reducing their harmful impacts. In this study, we identified a manganese peroxidase from Punctularia strigosozonata (PsMnp) through protein similarity analysis, which has the capability to degrade four AFs (AFB1, AFB2, AFG1, and AFG2) simultaneously. The gene encoding this enzyme was subject to codon optimization, followed by cold shock induction expression using the pColdII vector, leading to the soluble expression of manganese peroxidase (Mnp) in Escherichia coli. This study tackled the problem of inclusion body formation that often occurs during Mnp expression in E. coli. After optimizing the degradation conditions, the degradation rates for AFB1, AFB2, AFG1, and AFG2 were 87.9, 72.8, 77.3, and 85.6%, respectively. Molecular docking and molecular dynamics simulations indicated that PsMnp facilitated the degradation of AFs through hydrophobic and polar interactions among various amino acid residues. This research offers novel insights into the rapid discovery of enzymes capable of degrading AFs and establishes a theoretical foundation for the efficient expression of mycotoxin detoxification enzymes.
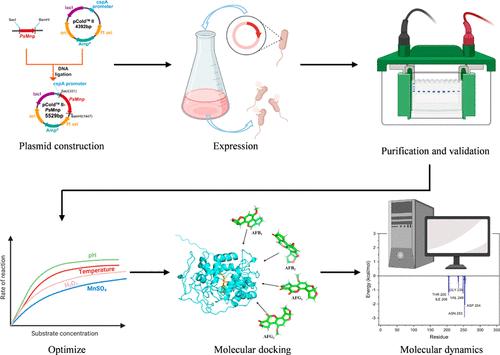
同时降解黄曲霉毒素AFB1、AFB2、AFG1和AFG2的锰过氧化物酶的高效表达和活性优化
黄曲霉毒素是一种臭名昭著的真菌毒素,对人类和动物健康构成重大风险,因此生物降解极为重要,因为它们为管理和减少其有害影响提供了一种有希望的方法。在本研究中,我们通过蛋白质相似性分析,从刺状藻(Punctularia strigosozonata, PsMnp)中鉴定出一种锰过氧化物酶,该酶具有同时降解AFB1、AFB2、AFG1和AFG2四种AFs的能力。对编码该酶的基因进行密码子优化,然后利用pColdII载体进行冷休克诱导表达,使锰过氧化物酶(Mnp)在大肠杆菌中可溶表达。本研究解决了在大肠杆菌中Mnp表达过程中经常发生的包涵体形成问题。优化降解条件后,AFB1、AFB2、AFG1和AFG2的降解率分别为87.9、72.8、77.3和85.6%。分子对接和分子动力学模拟表明,PsMnp通过各种氨基酸残基之间的疏水和极性相互作用促进了AFs的降解。本研究为快速发现能够降解AFs的酶提供了新的见解,并为真菌毒素解毒酶的高效表达奠定了理论基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
9.90
自引率
8.20%
发文量
1375
审稿时长
2.3 months
期刊介绍:
The Journal of Agricultural and Food Chemistry publishes high-quality, cutting edge original research representing complete studies and research advances dealing with the chemistry and biochemistry of agriculture and food. The Journal also encourages papers with chemistry and/or biochemistry as a major component combined with biological/sensory/nutritional/toxicological evaluation related to agriculture and/or food.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


