Recombinant Fungal Aspartic Endopeptidases: Insights into Protein Hydrolysis and Combined Effect with Pepsin for Animal Feed Application
IF 5.7
1区 农林科学
Q1 AGRICULTURE, MULTIDISCIPLINARY
引用次数: 0
Abstract
Protein hydrolysis under acidic conditions can improve the product quality, nutrient availability, and cost efficiency, particularly when neutral or alkaline enzymes are ineffective. Six fungal aspartic endopeptidases (FAPs) were recombinantly expressed as active enzymes in Komagataella phaffi, with peak activity between 30–50 °C and pH 3.0–4.0. Despite FAP1 yielding a higher degree of hydrolysis for soy protein isolate (SPI) than FAP4, mass spectrometry analysis revealed similar cleavage preferences for the two peptidases. FAP1 and FAP4 experienced competitive product inhibition (Ki: 2.8 mg mL–1, Km: 3.2 mg mL–1 for FAP1 and Ki: 9.67 mg mL–1, Km: 6.58 mg mL–1 for FAP4). These findings suggest that Ki and Km values, when studied in isolation, do not always predict a peptidase’s hydrolytic efficacy. Among the FAPs, FAP6 notably increased soluble protein content in animal feed by ∼3-fold. FAP1, when combined with pepsin, had a positive effect on the hydrolysis of SPI. These results underscore the potential of FAPs to hydrolyze proteins─specifically, animal feed proteins─in acidic environments.
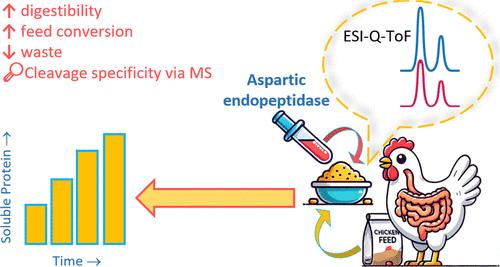
重组真菌天冬氨酸内肽酶:蛋白质水解及其与胃蛋白酶联合作用在饲料中的应用
在酸性条件下水解蛋白质可以提高产品质量、营养利用率和成本效率,特别是在中性或碱性酶无效的情况下。6个真菌天冬氨酸内肽酶(FAPs)在法菲Komagataella phaffi中重组表达,在30 ~ 50℃、pH 3.0 ~ 4.0之间具有最高活性。尽管FAP1对大豆分离蛋白(SPI)的水解程度高于FAP4,但质谱分析显示这两种肽酶的裂解偏好相似。FAP1和FAP4经历竞争性产物抑制(FAP1 Ki: 2.8 mg mL-1, Km: 3.2 mg mL-1, FAP4 Ki: 9.67 mg mL-1, Km: 6.58 mg mL-1)。这些发现表明,单独研究Ki和Km值时,并不总是预测肽酶的水解功效。在FAPs中,FAP6显著提高了动物饲料中可溶性蛋白含量约3倍。当FAP1与胃蛋白酶结合时,对SPI的水解有积极的影响。这些结果强调了FAPs在酸性环境中水解蛋白质(特别是动物饲料蛋白)的潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
9.90
自引率
8.20%
发文量
1375
审稿时长
2.3 months
期刊介绍:
The Journal of Agricultural and Food Chemistry publishes high-quality, cutting edge original research representing complete studies and research advances dealing with the chemistry and biochemistry of agriculture and food. The Journal also encourages papers with chemistry and/or biochemistry as a major component combined with biological/sensory/nutritional/toxicological evaluation related to agriculture and/or food.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


