Ancestral Nitrilase Mining and Semi-Rational Engineering for Enhanced Thermal Stability in Rapeseed Meals-Derived Nitriles Degradation
IF 5.7
1区 农林科学
Q1 AGRICULTURE, MULTIDISCIPLINARY
引用次数: 0
Abstract
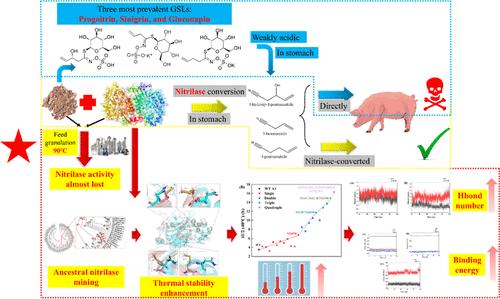
Rapeseed meal (RSM), a protein-rich byproduct, holds potential as a high-quality animal feed, but nitrile compounds derived from glucosinolates (GSLs) in RSM pose a toxicity risk. Nitrilases, enzymes that hydrolyze toxic nitriles to carboxylic acids, offer a potential solution for detoxification. However, the low thermal stability of nitrilases restricts their industrial applicability. We herein identified eight ancestral nitrilases through sequence-based mining using 6803NIT as a probe enzyme. Among these, ancestral enzyme A1 exhibited the highest specific activity (58.3 U/mg) and half-life (t1/2 = 3.5 h at 40 °C). To enhance thermal stability, we engineered a quadruple mutant A1M_4C, which exhibited a 4.7-fold increase in half-life (t1/2 = 16.3 h) and a 2-fold increase in specific activity (118.5 U/mg). Kinetic analysis revealed a reduction in Km from 14.9 to 10.5 mM and an increase in kcat/Km from 1.9 to 4.37 s–1·mM–1. Mechanistic studies indicated that enhanced stability in A1M_4C was due to increased hydrogen bonding and stronger amino acid interactions. Simulated feed pelletization at 90 °C for 2 min showed that A1M_4C acquired a 22.2-fold improvement toward nitriles degradation over wild-type A1. These findings demonstrate the potential of ancestral enzyme mining to develop thermostable nitrilases for industrial feed applications.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
9.90
自引率
8.20%
发文量
1375
审稿时长
2.3 months
期刊介绍:
The Journal of Agricultural and Food Chemistry publishes high-quality, cutting edge original research representing complete studies and research advances dealing with the chemistry and biochemistry of agriculture and food. The Journal also encourages papers with chemistry and/or biochemistry as a major component combined with biological/sensory/nutritional/toxicological evaluation related to agriculture and/or food.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


