Untargeted Spatial Metabolomics and Spatial Proteomics on the Same Tissue Section
IF 6.7
1区 化学
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
Abstract
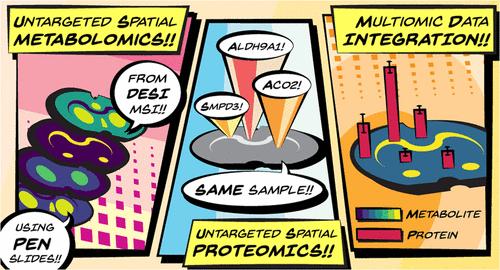
An increasing number of spatial multiomic workflows have recently been developed. Some of these approaches have leveraged initial mass spectrometry imaging (MSI)-based spatial metabolomics to inform the region of interest (ROI) selection for downstream spatial proteomics. However, these workflows have been limited by varied substrate requirements between modalities or have required analyzing serial sections (i.e., one section per modality). To mitigate these issues, we present a new multiomic workflow that uses desorption electrospray ionization (DESI)-MSI to identify representative spatial metabolite patterns on-tissue prior to spatial proteomic analyses on the same tissue section. This workflow is demonstrated here with a model mammalian tissue (coronal rat brain section) mounted on a poly(ethylene naphthalate)-membrane slide. Initial DESI-MSI resulted in 160 annotations (SwissLipids) within the METASPACE platform (≤20% false discovery rate). A segmentation map from the annotated ion images informed the downstream ROI selection for spatial proteomics characterization from the same sample. The unspecific substrate requirements and minimal sample disruption inherent to DESI-MSI allowed for an optimized, downstream spatial proteomics assay, resulting in 3888 ± 240 to 4717 ± 48 proteins being confidently directed per ROI (200 μm × 200 μm). Finally, we demonstrate the integration of multiomic information, where we found ceramide localization to be correlated with SMPD3 abundance (ceramide synthesis protein), and we also utilized protein abundance to resolve metabolite isomeric ambiguity. Overall, the integration of DESI-MSI into the multiomic workflow allows for complementary spatial- and molecular-level information to be achieved from optimized implementations of each MS assay inherent to the workflow itself.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Analytical Chemistry
化学-分析化学
CiteScore
12.10
自引率
12.20%
发文量
1949
审稿时长
1.4 months
期刊介绍:
Analytical Chemistry, a peer-reviewed research journal, focuses on disseminating new and original knowledge across all branches of analytical chemistry. Fundamental articles may explore general principles of chemical measurement science and need not directly address existing or potential analytical methodology. They can be entirely theoretical or report experimental results. Contributions may cover various phases of analytical operations, including sampling, bioanalysis, electrochemistry, mass spectrometry, microscale and nanoscale systems, environmental analysis, separations, spectroscopy, chemical reactions and selectivity, instrumentation, imaging, surface analysis, and data processing. Papers discussing known analytical methods should present a significant, original application of the method, a notable improvement, or results on an important analyte.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


