Determination of the pKa and Concentration of NMR-Invisible Molecules and Sites Using NMR Spectroscopy
IF 6.7
1区 化学
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
Abstract
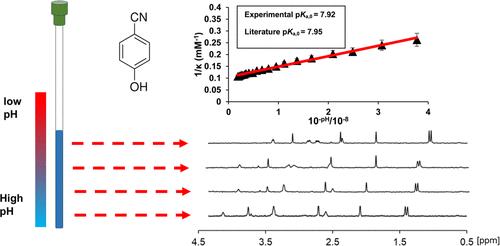
NMR spectroscopy is a very powerful tool for measuring the dissociation constants (pKa) of molecules, requiring smaller quantities of samples of lower purity relative to potentiometric or conductometric methods. However, current approaches are generally limited to those molecules possessing favorable pH-dependent NMR properties. Typically, a series of 1D experiments at varying pH are performed, and the pKa is obtained by fitting the observed chemical shift of the analyte as a function of pH using nonlinear routines. However, the majority of polymers, biomolecules, and inorganic species do not present favorable NMR resonances. Either the resonances are not observable or too broad, or the unambiguous interpretation of the NMR data is impossible without resorting to complex 2D experiments due to spectral overlap. To overcome these fundamental limitations, we present a method to obtain the pKa values and concentrations of acidic species without their direct observation by NMR. We instead determine the quantity of acidic protons removed from the species along a concentration gradient of an organic base in a single 1H chemical shift imaging experiment that can be run under automation. The pKa values are determined via simple linear plots, avoiding complex and potentially unreliable nonlinear fitting routines.
核磁共振波谱法测定核磁共振不可见分子和位点的pKa和浓度
核磁共振波谱是测量分子解离常数(pKa)的一种非常强大的工具,相对于电位法或电导法,它需要的样品数量更少,纯度更低。然而,目前的方法通常仅限于那些具有有利的ph依赖核磁共振性质的分子。通常,在不同的pH下进行一系列一维实验,并通过使用非线性程序将观察到的分析物的化学位移拟合为pH的函数来获得pKa。然而,大多数聚合物、生物分子和无机物不表现出良好的核磁共振。要么是共振不可观察或太广泛,要么是由于光谱重叠而不诉诸复杂的二维实验,不可能对核磁共振数据进行明确的解释。为了克服这些基本的限制,我们提出了一种不用核磁共振直接观察酸性物质的pKa值和浓度的方法。相反,我们在一个可以在自动化下运行的单1H化学位移成像实验中,沿着有机碱的浓度梯度确定从物种中去除的酸性质子的数量。pKa值通过简单的线性图确定,避免了复杂和可能不可靠的非线性拟合程序。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Analytical Chemistry
化学-分析化学
CiteScore
12.10
自引率
12.20%
发文量
1949
审稿时长
1.4 months
期刊介绍:
Analytical Chemistry, a peer-reviewed research journal, focuses on disseminating new and original knowledge across all branches of analytical chemistry. Fundamental articles may explore general principles of chemical measurement science and need not directly address existing or potential analytical methodology. They can be entirely theoretical or report experimental results. Contributions may cover various phases of analytical operations, including sampling, bioanalysis, electrochemistry, mass spectrometry, microscale and nanoscale systems, environmental analysis, separations, spectroscopy, chemical reactions and selectivity, instrumentation, imaging, surface analysis, and data processing. Papers discussing known analytical methods should present a significant, original application of the method, a notable improvement, or results on an important analyte.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


