Ruminant Trans Fatty Acid Intake Modulates Inflammation Pathways in the Adipose Tissue Transcriptome of C57BL/6 Mice
Abstract
Scope
The study aims to analyze transcriptomic profiles in adipose tissues postconsumption of elaidic acid (EA; trans-18:1n-9) and trans-palmitoleic acid (TPA; trans-16:1n-7), elucidating their different effects on inflammation and glucose metabolism.
Methods and results
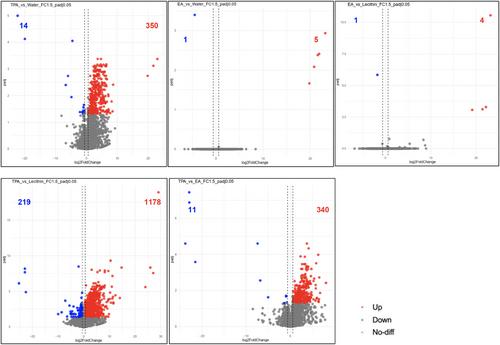
Twenty C57BL/6 mice are divided into four groups. Each group receives one of the following formulations in drinking water: lecithin nanovesicles, nanovesicles containing either lecithin with EA or TPA (86:14 w/w), or water (control) for 28 days with a regular fat diet (18% calories from fat). Total RNA is extracted, and paired-end sequencing is performed. TPA intake alters the expression of 351 genes compared to EA intake, including 11 downregulated and 340 upregulated genes (fold change [FC] >1.5, p < 0.05). TPA compares to EA upregulated: Slc5a8, Lcn2, Csf3, Scube1, Mapk13, Bdkrb2, Ctla2a, Slc2a1, Oas3, Cx3cl1, Oas2, Nlrp6, Pycard, Cyba, Ddr1, and Prkab1 and downregulated Fas gene. These genes are related to the NOD-like receptor, lipid and atherosclerosis, IL-17 signaling, TNF, nonalcoholic fatty liver disease, cytokine–cytokine receptor interaction, adipocytokine, glucagon, insulin resistance, and inflammatory mediator regulation of TRP channels signaling.
Conclusion
TPA intake has a distinct impact on the regulation of inflammation and diabetes-related pathways in adipose tissue compared to EA.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


