Epigenetic regulators of clonal hematopoiesis control CD8 T cell stemness during immunotherapy
IF 45.8
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
Epigenetic reinforcement of T cell exhaustion is known to be a major barrier limiting T cell responses during immunotherapy. However, the core epigenetic regulators restricting antitumor immunity during prolonged antigen exposure are not clear. We investigated three commonly mutated epigenetic regulators that promote clonal hematopoiesis to determine whether they affect T cell stemness and response to checkpoint blockade immunotherapy. CD8 T cells lacking Dnmt3a, Tet2, or Asxl1 preserved a progenitor-exhausted (Tpex) population for more than 1 year during chronic antigen exposure without undergoing malignant transformation. Asxl1 controlled the self-renewal capacity of T cells and reduced CD8 T cell differentiation through H2AK119 ubiquitination and epigenetic modification of the polycomb group–repressive deubiquitinase pathway. Asxl1-deficient T cells synergized with anti–PD-L1 immunotherapy to improve tumor control in experimental models and conferred a survival advantage to mutated T cells from treated patients.
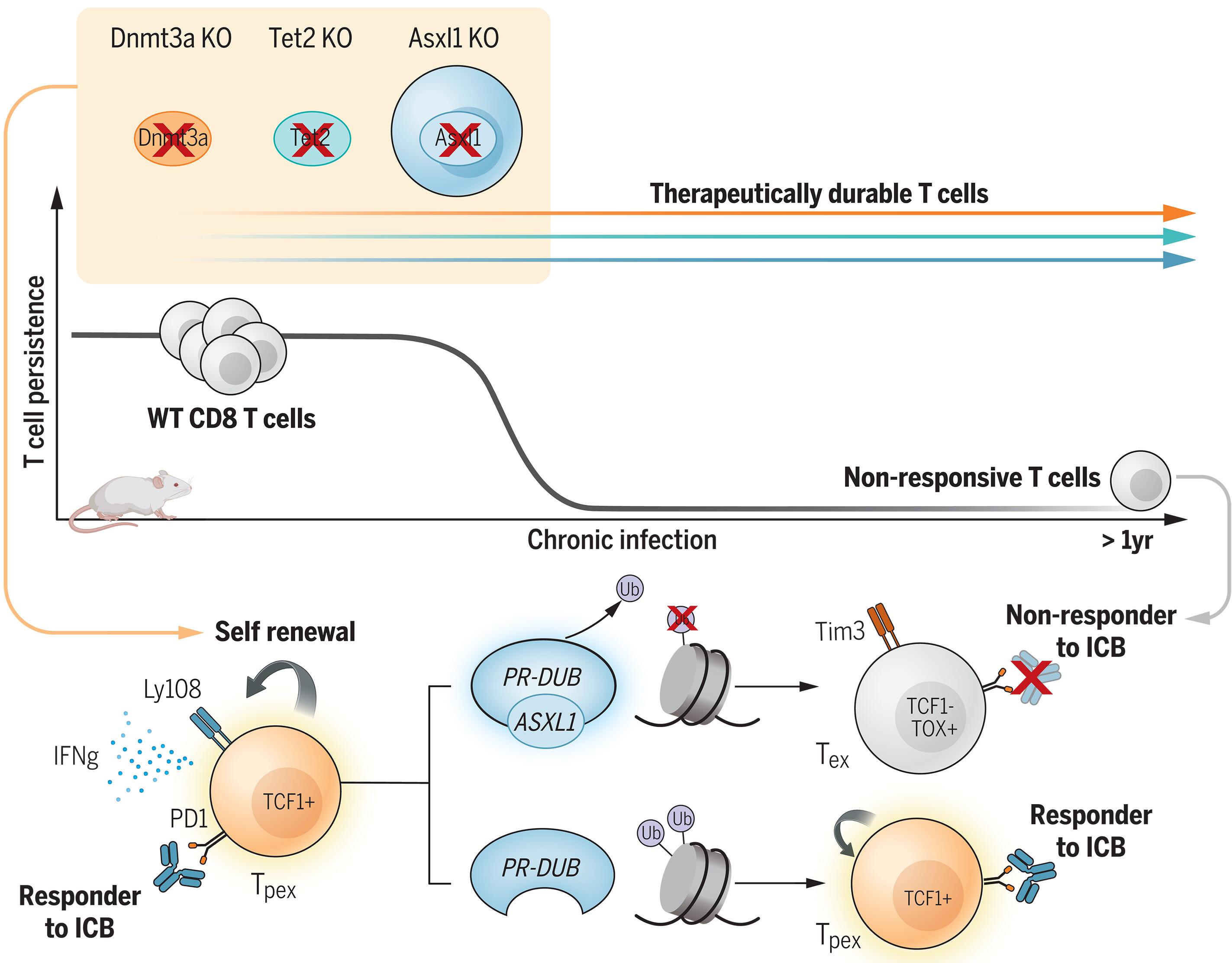
克隆造血的表观遗传调节因子在免疫疗法中控制 CD8 T 细胞干性。
众所周知,T细胞衰竭的表观遗传强化是限制免疫疗法中T细胞反应的主要障碍。然而,在长期抗原暴露过程中限制抗肿瘤免疫的核心表观遗传调节因子尚不清楚。我们研究了三种促进克隆造血的常见突变表观遗传调节因子,以确定它们是否会影响T细胞的干性和对检查点阻断免疫疗法的反应。缺乏Dnmt3a、Tet2或Asxl1的CD8 T细胞在慢性抗原暴露期间保留了祖细胞耗竭(Tpex)群体1年以上,而没有发生恶性转化。Asxl1控制了T细胞的自我更新能力,并通过H2AK119泛素化和多聚酶组抑制性去泛素化酶途径的表观遗传修饰减少了CD8 T细胞的分化。Asxl1缺陷T细胞与抗PD-L1免疫疗法协同作用,改善了实验模型中的肿瘤控制,并使接受治疗的患者的突变T细胞具有生存优势。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Science
综合性期刊-综合性期刊
CiteScore
61.10
自引率
0.90%
发文量
0
审稿时长
2.1 months
期刊介绍:
Science is a leading outlet for scientific news, commentary, and cutting-edge research. Through its print and online incarnations, Science reaches an estimated worldwide readership of more than one million. Science’s authorship is global too, and its articles consistently rank among the world's most cited research.
Science serves as a forum for discussion of important issues related to the advancement of science by publishing material on which a consensus has been reached as well as including the presentation of minority or conflicting points of view. Accordingly, all articles published in Science—including editorials, news and comment, and book reviews—are signed and reflect the individual views of the authors and not official points of view adopted by AAAS or the institutions with which the authors are affiliated.
Science seeks to publish those papers that are most influential in their fields or across fields and that will significantly advance scientific understanding. Selected papers should present novel and broadly important data, syntheses, or concepts. They should merit recognition by the wider scientific community and general public provided by publication in Science, beyond that provided by specialty journals. Science welcomes submissions from all fields of science and from any source. The editors are committed to the prompt evaluation and publication of submitted papers while upholding high standards that support reproducibility of published research. Science is published weekly; selected papers are published online ahead of print.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


