Investigating the interaction mechanisms between arachin and resveratrol: Utilizing multi-spectroscopy and computational chemistry
IF 8.5
1区 农林科学
Q1 CHEMISTRY, APPLIED
引用次数: 0
Abstract
Arachin (ARA) and resveratrol (RES) are the primary protein and bioactive compound in peanuts and their processed products. However, the mechanism of interaction between these two substances remained unclear. To investigate protein structural changes, conformational variations, and molecular mechanisms in the interaction between them, multispectral analysis and computational chemistry methods were employed. Experimental results confirmed that RES quenched ARA's intrinsic fluorescence through static quenching, indicating their interaction. Thermodynamic analysis revealed the interaction between them was endothermic, spontaneous, and primarily hydrophobic. Molecular dynamics (MD) simulations highlighted strong affinity between RES and ARA, with key amino acids (His425, Val426, Phe405, and Phe464) facilitating their interaction. RES binding increased stability without significant protein conformational changes. The independent gradient model based on Hirshfeld partition (IGMH) validated their interaction, emphasizing van der Waals (VDW) interactions and hydrogen bonds (H-bonds) as crucial for stable binding. This research lays a theoretical foundation for potential applications of ARA-RES complex products in the food industry.
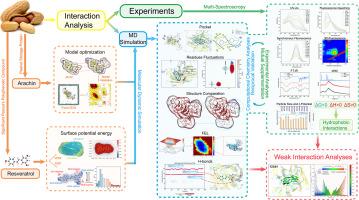
研究花青素和白藜芦醇之间的相互作用机制:利用多光谱学和计算化学
花生素(ARA)和白藜芦醇(RES)是花生及其加工产品中的主要蛋白质和生物活性化合物。然而,这两种物质之间的相互作用机制仍不清楚。为了研究这两种物质相互作用过程中的蛋白质结构变化、构象变化和分子机制,研究人员采用了多光谱分析和计算化学方法。实验结果证实,RES 通过静态淬灭作用淬灭了 ARA 的本征荧光,表明它们之间存在相互作用。热力学分析表明它们之间的相互作用是内热、自发和主要疏水的。分子动力学(MD)模拟突显了 RES 和 ARA 之间的强亲和力,关键氨基酸(His425、Val426、Phe405 和 Phe464)促进了它们之间的相互作用。RES 的结合提高了蛋白质的稳定性,而蛋白质的构象却没有发生重大变化。基于 Hirshfeld 分区的独立梯度模型(IGMH)验证了它们之间的相互作用,强调范德华(VDW)相互作用和氢键(H-bonds)是稳定结合的关键。这项研究为 ARA-RES 复合物产品在食品工业中的潜在应用奠定了理论基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Food Chemistry
工程技术-食品科技
CiteScore
16.30
自引率
10.20%
发文量
3130
审稿时长
122 days
期刊介绍:
Food Chemistry publishes original research papers dealing with the advancement of the chemistry and biochemistry of foods or the analytical methods/ approach used. All papers should focus on the novelty of the research carried out.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


