免疫信息学指导为免疫功能受损患者设计和评估基于蛋白质和信使核糖核酸的新型隐球菌疫苗。
IF 2.8
Q3 Biochemistry, Genetics and Molecular Biology
Journal of Genetic Engineering and Biotechnology
Pub Date : 2023-12-01
DOI:10.1186/s43141-023-00560-3
引用次数: 0
摘要
背景:新型隐球菌是一种真菌病原体,可在因HIV/AIDS(人类免疫缺陷病毒/获得性免疫缺陷综合征)、肝硬化和移植而导致免疫系统受损的个体中引起严重的脑膜脑炎。甘露糖蛋白(MP)是新生隐球菌胶囊中的糖蛋白,通过介导与肺细胞的粘附和通过细胞因子诱导和吞噬作用影响调节免疫反应,对毒力产生至关重要的影响。因此,研制一种能够产生靶向抗体以对抗感染和预防真菌疾病的疫苗至关重要。结果:本研究旨在通过生物信息学方法,针对新生隐球菌MP中发现的T和B细胞表位,创造一种独特、稳定、安全的疫苗。基于毒性、免疫原性和抗原性,这项研究预测了新的T细胞(GNPVGGNVT、NPVGGNVTT、QTSYARLLS、TSVGNGIAS、WVMPGDYTN、AAATGSSSSGSTGSG、GSTGSGSGAAAGST、SGSTGSGSAAAGS、SSGSTGSGAAAG和SSSGSTGSGGSGSGSGSAAA)和B细胞GPTNPSEPTLGTAI、GNPVGGNVTTNATGSD和NSTAAATNSSGGASA)多表位疫苗的表位,并构建了具有潜在免疫原性特性的疫苗亚单位。本研究使用四种连接体(AAY、GPGPG、KK和EAAAK连接体)连接表位和佐剂。在构建疫苗后,它面临着受体对接和模拟分析。随后,将该疫苗克隆到大肠杆菌pET-28a载体中( +) 通过使用SnapGene工具进行表达的连接过程,这证实了显著的免疫反应。为了评估构建的疫苗的特性,使用了多种计算工具。基于MP序列,该工具评估了抗原性、免疫原性、细胞因子诱导能力、致敏性、毒性、人群覆盖率和溶解度。结论:最终,研究结果揭示了一种有前景的多表位疫苗,它是解决全球新生隐球菌感染的潜在候选疫苗,特别是在免疫功能低下的患者中。然而,需要进行更多的体外和体内研究来验证其安全性和有效性。本文章由计算机程序翻译,如有差异,请以英文原文为准。

Immunoinformatic-guided designing and evaluating protein and mRNA-based vaccines against Cryptococcus neoformans for immunocompromised patients
Background
Cryptococcus neoformans is a fungal pathogen that can cause serious meningoencephalitis in individuals with compromised immune systems due to HIV/AIDS (human immunodeficiency virus/acquired immunodeficiency syndrome), liver cirrhosis, and transplantation. Mannoproteins (MPs), glycoproteins in the C. neoformans capsule, crucially impact virulence by mediating adhesion to lung cells and modulating immune response via cytokine induction and phagocytosis influence. Therefore, creating a vaccine that can generate targeted antibodies to fight infection and prevent fungal illnesses is essential.
Results
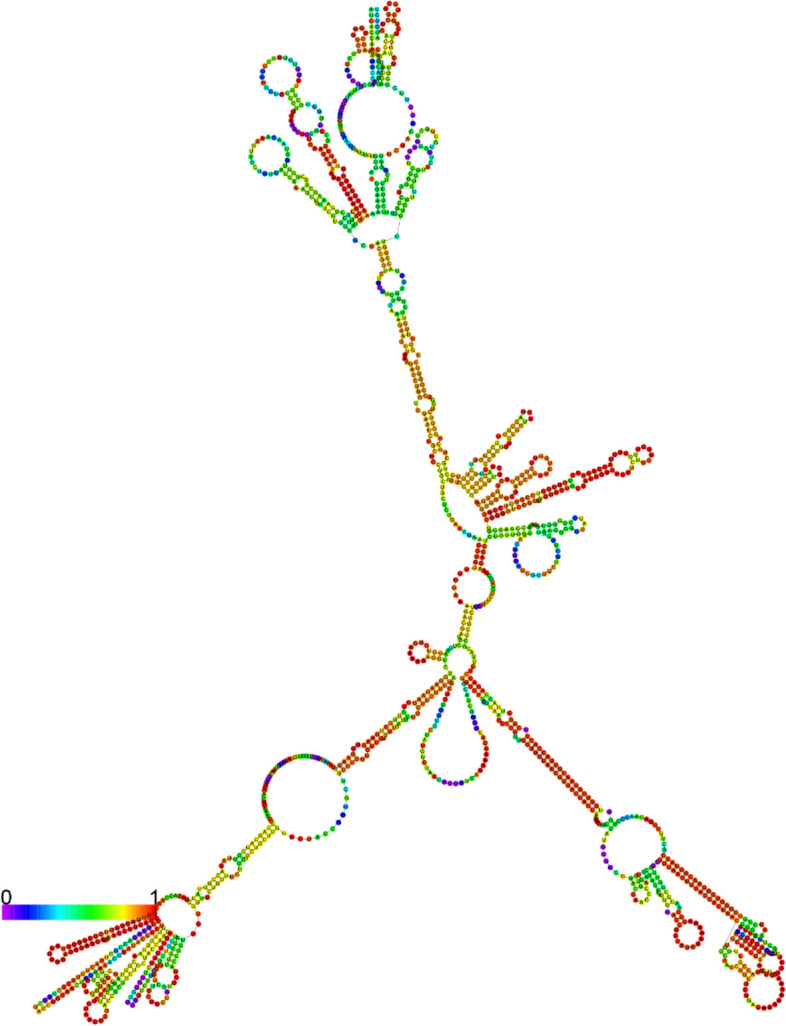
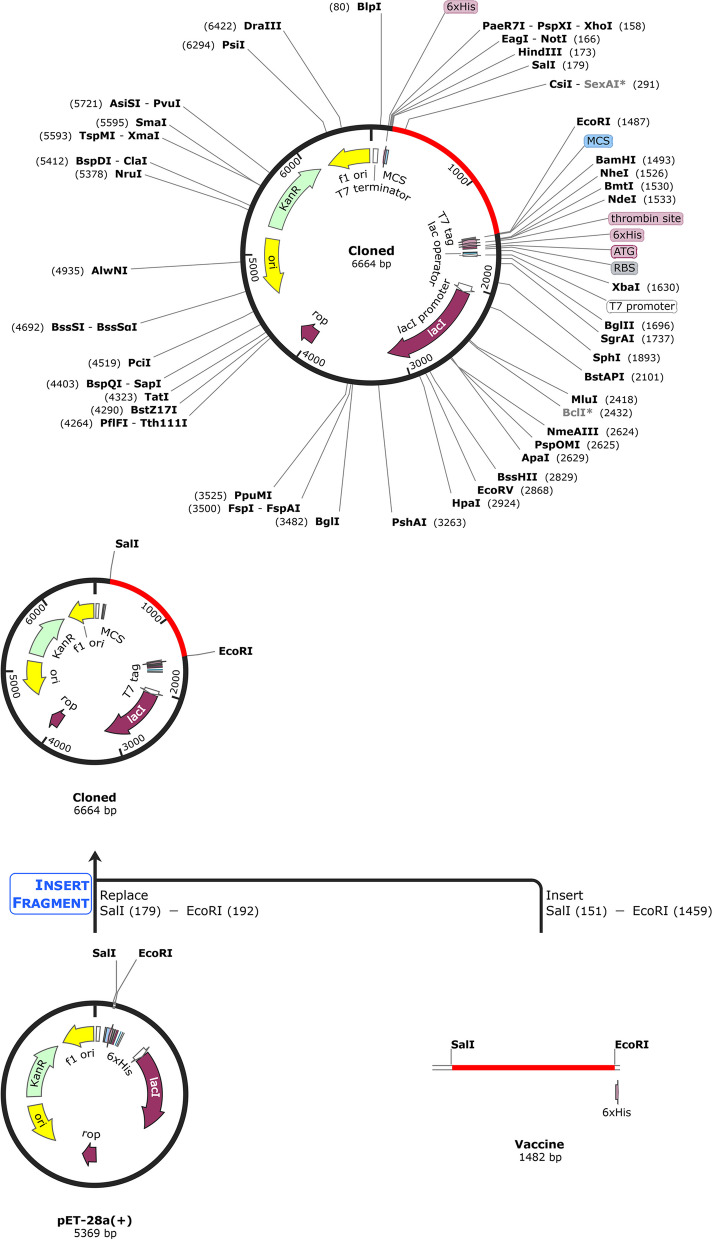
This research aims to create a unique, stable, and safe vaccine through bioinformatics methodologies, aiming at epitopes of T and B cells found in the MP of C. neoformans. Based on toxicity, immunogenicity, and antigenicity, this research predicted novel T cells (GNPVGGNVT, NPVGGNVTT, QTSYARLLS, TSVGNGIAS, WVMPGDYTN, AAATGSSSSGSTGSG, GSTGSGSGSAAAGST, SGSTGSGSGSAAAGS, SSGSTGSGSGSAAAG, and SSSGSTGSGSGSAAA) and B cell (ANGSTSTFQQRYTGTYTNGDGSLGTWTQGETVTPQTAYSTPATSNCKTYTSVGNGIASLALSNAGSNSTAAATNSSSGGASAAATGSSSSGSTGSGSGSAAAGSTAAASSSGDSSSSTSAAMSNGI, HGATGLGNPVGGNVTT, TMGPTNPSEPTLGTAI, GNPVGGNVTTNATGSD, and NSTAAATNSSSGGASA) epitopes for a multiple-epitope vaccine and constructed a vaccine subunit with potential immunogenic properties. The present study used four linkers (AAY, GPGPG, KK, and EAAAK linkers) to connect the epitopes and adjuvant. After constructing the vaccine, it was confronted with receptor docking and simulation analysis. Subsequently, the vaccine was cloned into the vector of Escherichia coli pET-28a (+) by ligation process for the expression using the SnapGene tool, which confirmed a significant immune response. To assess the constructed vaccine’s properties, multiple computational tools were employed. Based on the MP sequence, the tools evaluated the antigenicity, immunogenicity, cytokine-inducing capacity, allergenicity, toxicity, population coverage, and solubility.
Conclusion
Eventually, the results revealed a promising multi-epitope vaccine as a potential candidate for addressing global C. neoformans infection, particularly in immunocompromised patients. Yet, additional in vitro and in vivo investigations are necessary to validate its safety and effectiveness.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Genetic Engineering and Biotechnology
Biochemistry, Genetics and Molecular Biology-Biotechnology
CiteScore
5.70
自引率
5.70%
发文量
159
审稿时长
16 weeks
期刊介绍:
Journal of genetic engineering and biotechnology is devoted to rapid publication of full-length research papers that leads to significant contribution in advancing knowledge in genetic engineering and biotechnology and provide novel perspectives in this research area. JGEB includes all major themes related to genetic engineering and recombinant DNA. The area of interest of JGEB includes but not restricted to: •Plant genetics •Animal genetics •Bacterial enzymes •Agricultural Biotechnology, •Biochemistry, •Biophysics, •Bioinformatics, •Environmental Biotechnology, •Industrial Biotechnology, •Microbial biotechnology, •Medical Biotechnology, •Bioenergy, Biosafety, •Biosecurity, •Bioethics, •GMOS, •Genomic, •Proteomic JGEB accepts
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


