叔n -苯基cf3 -亚氨基丙基醇的加氢卤化反应
IF 1.7
4区 化学
Q3 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
摘要
通过易得的叔n-苯基cf3 -亚氨基丙基醇与氢卤酸(HCl, HBr, HI)反应,建立了制备4-卤-5,5-烷基/芳基- n-苯基-2-(三氟甲基)-2,5-二氢呋喃-2胺的简便方法。得到的4-halo-2,5-二氢呋喃很容易发生反应,表明它们在合成各种含cf3的化合物中具有潜在的用途。本文章由计算机程序翻译,如有差异,请以英文原文为准。
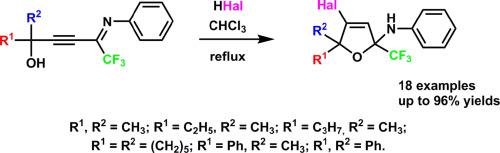
Hydrohalogenation of tertiary N-phenyl CF3-iminopropargylic alcohols
A facile approach towards 4-halo-5,5-alkyl/aryl-N-phenyl-2-(trifluoromethyl)-2,5-dihydrofuran-2-amines by the reaction of readily available tertiary N-phenyl CF3-iminopropargylic alcohols with hydrohalic acids (HCl, HBr, HI) was developed. The obtained 4-halo-2,5-dihydrofurans were shown to readily undergo the reactions demonstrating their potential utility in the synthesis of various CF3-containing compounds.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Fluorine Chemistry
化学-无机化学与核化学
CiteScore
3.80
自引率
10.50%
发文量
99
审稿时长
33 days
期刊介绍:
The Journal of Fluorine Chemistry contains reviews, original papers and short communications. The journal covers all aspects of pure and applied research on the chemistry as well as on the applications of fluorine, and of compounds or materials where fluorine exercises significant effects. This can include all chemistry research areas (inorganic, organic, organometallic, macromolecular and physical chemistry) but also includes papers on biological/biochemical related aspects of Fluorine chemistry as well as medicinal, agrochemical and pharmacological research. The Journal of Fluorine Chemistry also publishes environmental and industrial papers dealing with aspects of Fluorine chemistry on energy and material sciences. Preparative and physico-chemical investigations as well as theoretical, structural and mechanistic aspects are covered. The Journal, however, does not accept work of purely routine nature.
For reviews and special issues on particular topics of fluorine chemistry or from selected symposia, please contact the Regional Editors for further details.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


