Tirzepatide在超重或肥胖成年人强化生活方式干预后的疗效:SURMOUNT-3 3期试验。
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 3
摘要
替西帕肽是一种葡萄糖依赖性促胰岛素多肽和胰高血糖素样肽-1受体激动剂,在成功的强化生活方式干预后对减肥的影响尚不清楚。这项双盲安慰剂对照试验将体重指数为1:1的成年人随机分组 ≥30或≥27 kg/m2和至少一种与肥胖相关的并发症(不包括糖尿病),在12周的强化生活方式干预后体重减轻≥5.0%,达到替西帕肽的最大耐受剂量(10或15 mg)或安慰剂,每周一次,持续72 周(n = 579)。在意向治疗人群中,无论治疗依从性如何,治疗方案都会评估效果。随机分组至一周的额外平均重量百分比变化的共同主要终点 72例患者的替西帕肽变化为-18.4%(标准误差(s.e.0.7),安慰剂变化为2.5%(s.e.1.0)(估计治疗差异-20.8个百分点(95%置信区间(CI)-23.2%,-18.5%;P本文章由计算机程序翻译,如有差异,请以英文原文为准。
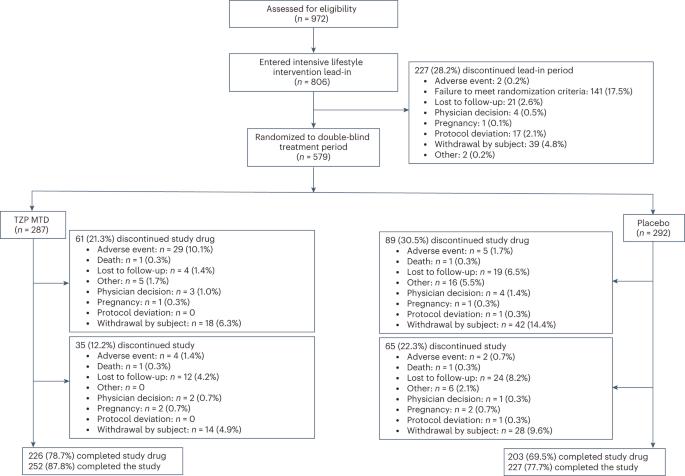
Tirzepatide after intensive lifestyle intervention in adults with overweight or obesity: the SURMOUNT-3 phase 3 trial
The effects of tirzepatide, a glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist, on weight reduction after successful intensive lifestyle intervention are unknown. This double-blind, placebo-controlled trial randomized (1:1) adults with body mass index ≥30 or ≥27 kg/m2 and at least one obesity-related complication (excluding diabetes), who achieved ≥5.0% weight reduction after a 12-week intensive lifestyle intervention, to tirzepatide maximum tolerated dose (10 or 15 mg) or placebo once weekly for 72 weeks (n = 579). The treatment regimen estimand assessed effects regardless of treatment adherence in the intention-to-treat population. The coprimary endpoint of additional mean per cent weight change from randomization to week 72 was met with changes of −18.4% (standard error (s.e.) 0.7) with tirzepatide and 2.5% (s.e. 1.0) with placebo (estimated treatment difference −20.8 percentage points (95% confidence interval (CI) −23.2%, −18.5%; P < 0.001). The coprimary endpoint of the percentage of participants achieving additional weight reduction ≥5% was met with 87.5% (s.e. 2.2) with tirzepatide and 16.5% (s.e. 3.0) with placebo achieving this threshold (odds ratio 34.6%; 95% CI 19.2%, 62.6%; P < 0.001). The most common adverse events with tirzepatide were gastrointestinal, with most being mild to moderate in severity. Tirzepatide provided substantial additional reduction in body weight in participants who had achieved ≥5.0% weight reduction with intensive lifestyle intervention. ClinicalTrials.gov registration: NCT04657016 . In the SURMOUNT-3 trial, once-weekly treatment with tirzepatide was demonstrated to result in clinically meaningful additional weight loss in adults with overweight or obesity following initial successful weight loss of at least 5% body weight with intensive lifestyle intervention.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


