在水中或与水一起进行的羧酸的简单、绿色和官能团耐受还原
IF 9.3
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
羧酸在初始转化为相应的s -2-吡啶基硫酯时,在温和的条件下可以容易地还原为醛或醇。在用市售的空气稳定的镍预催化剂和硅烷作为化学计量还原剂处理后,醛以中等到良好的收率形成。或者,在用硼氢化钠处理后,酸的1锅转化为它们的硫酯衍生物可以接着还原为醇。各种各样的起始材料,从高功能化酸到从默克信息库的衍生物,都可以使用这些绿色反应介质进行转化。本文章由计算机程序翻译,如有差异,请以英文原文为准。
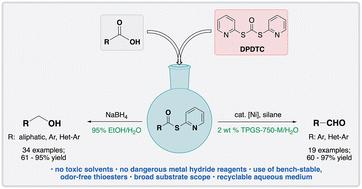
Facile, green, and functional group-tolerant reductions of carboxylic acids…in, or with, water†
Facile reductions of carboxylic acids to aldehydes or alcohols can be effected under mild conditions upon initial conversion to their corresponding S-2-pyridyl thioesters. Upon treatment with a commercially available and air-stable nickel pre-catalyst and silane as a stoichiometric reductant, aldehydes are formed in moderate to good yields. Alternatively, the 1-pot conversion of acids to their thioester derivatives can be followed by reduction to the alcohol upon treatment with sodium borohydride. A variety of starting materials ranging from highly functionalized acids to educts from the Merck informer library can be transformed using these green reaction media.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Green Chemistry
化学-化学综合
CiteScore
16.10
自引率
7.10%
发文量
677
审稿时长
1.4 months
期刊介绍:
Green Chemistry is a journal that provides a unique forum for the publication of innovative research on the development of alternative green and sustainable technologies. The scope of Green Chemistry is based on the definition proposed by Anastas and Warner (Green Chemistry: Theory and Practice, P T Anastas and J C Warner, Oxford University Press, Oxford, 1998), which defines green chemistry as the utilisation of a set of principles that reduces or eliminates the use or generation of hazardous substances in the design, manufacture and application of chemical products. Green Chemistry aims to reduce the environmental impact of the chemical enterprise by developing a technology base that is inherently non-toxic to living things and the environment. The journal welcomes submissions on all aspects of research relating to this endeavor and publishes original and significant cutting-edge research that is likely to be of wide general appeal. For a work to be published, it must present a significant advance in green chemistry, including a comparison with existing methods and a demonstration of advantages over those methods.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


