内质网折叠蛋白反应中的信号整合
IF 81.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 5758
摘要
由于未折叠蛋白质具有毒性,它们在内质网中的积累会激活细胞应激反应。这种未折叠蛋白质反应会重塑分泌途径,以适应未折叠蛋白质的负荷;如果负荷无法承受,则会促进细胞死亡,以保护生物体。内质网(ER)通过激活细胞内信号转导通路(统称为 "未折叠蛋白质反应")来应对其腔内未折叠蛋白质的积累(ER 应激)。UPR 至少有三个不同的机制臂,它们共同调控着分泌途径中许多基因的表达,同时也影响着细胞命运以及蛋白质、氨基酸和脂质代谢的方方面面。UPR 的各个臂集成在一起,提供了一种重塑分泌装置的反应,并使细胞生理学符合 ER 应激的要求。本文章由计算机程序翻译,如有差异,请以英文原文为准。
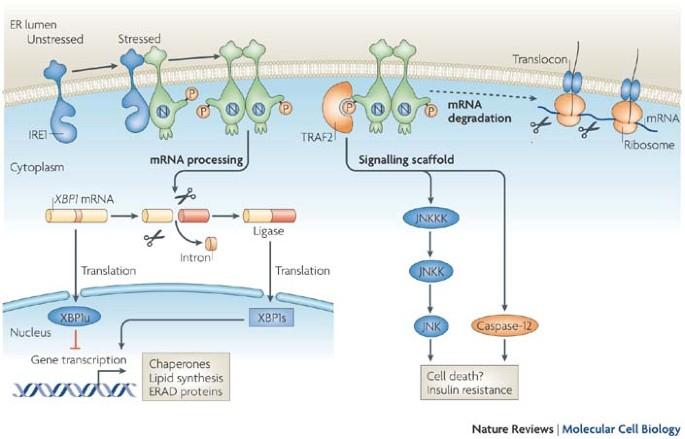
Signal integration in the endoplasmic reticulum unfolded protein response
Owing to the toxic potential of unfolded proteins, their accumulation in the endoplasmic reticulum activates a cellular stress response. This unfolded protein response remodels the secretory pathway to accommodate the load of unfolded proteins or, if the burden is insurmountable, promotes cell death to protect the organism. The endoplasmic reticulum (ER) responds to the accumulation of unfolded proteins in its lumen (ER stress) by activating intracellular signal transduction pathways — cumulatively called the unfolded protein response (UPR). Together, at least three mechanistically distinct arms of the UPR regulate the expression of numerous genes that function within the secretory pathway but also affect broad aspects of cell fate and the metabolism of proteins, amino acids and lipids. The arms of the UPR are integrated to provide a response that remodels the secretory apparatus and aligns cellular physiology to the demands imposed by ER stress.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
173.60
自引率
0.50%
发文量
118
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews Molecular Cell Biology is a prestigious journal that aims to be the primary source of reviews and commentaries for the scientific communities it serves. The journal strives to publish articles that are authoritative, accessible, and enriched with easily understandable figures, tables, and other display items. The goal is to provide an unparalleled service to authors, referees, and readers, and the journal works diligently to maximize the usefulness and impact of each article. Nature Reviews Molecular Cell Biology publishes a variety of article types, including Reviews, Perspectives, Comments, and Research Highlights, all of which are relevant to molecular and cell biologists. The journal's broad scope ensures that the articles it publishes reach the widest possible audience.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


