磷酸化激活Cdk:将生长信号与细胞周期控制联系起来。
IF 4.3
3区 生物学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
细胞根据外部因素和营养调节其增殖。这些输入必须到达细胞周期机制,以确保适当的细胞增殖。这篇综述的重点是有证据表明,周期蛋白依赖性激酶的t环域磷酸化可能是这些外部信号的一个关键和保守的通道。了解这种调节机制可以为真核细胞如何整合外部信息来决定是否分裂提供重要的见解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
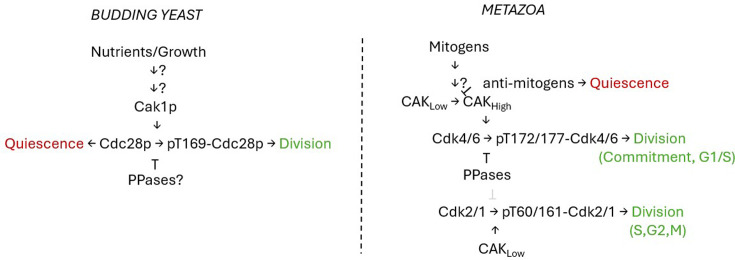
Cdk activation by phosphorylation: linking growth signals to cell cycle control.
Cells adjust their proliferation in response to extrinsic factors and nutrients. Such inputs must reach the cell cycle machinery to ensure proper cell proliferation. This minireview focuses on evidence suggesting that phosphorylating the T-loop domain of cyclin-dependent kinases may be a critical and conserved conduit for these external signals. Understanding this regulatory mechanism could provide crucial insights into how all eukaryotic cells integrate external information to decide whether or not to divide.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Biochemical Society transactions
生物-生化与分子生物学
CiteScore
7.80
自引率
0.00%
发文量
351
审稿时长
3-6 weeks
期刊介绍:
Biochemical Society Transactions is the reviews journal of the Biochemical Society. Publishing concise reviews written by experts in the field, providing a timely snapshot of the latest developments across all areas of the molecular and cellular biosciences.
Elevating our authors’ ideas and expertise, each review includes a perspectives section where authors offer comment on the latest advances, a glimpse of future challenges and highlighting the importance of associated research areas in far broader contexts.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


