放大到基因激活:雌激素受体α二聚化和DNA结合的高速原子力显微镜可视化
IF 16
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
雌激素受体α (ERα)在基因调控中起关键作用,特别是在雌激素反应性癌症中。然而,ERα的全长分子动力学结构尚不清楚。在这项研究中,我们使用高速原子力显微镜(HS-AFM)来观察在配体存在和不存在的情况下,ERα与雌激素反应元件(ERE)的相互作用。即使在没有雌激素的情况下,ERα也能与ERE结合,尽管配体的存在显著提高了结合的精度和稳定性。我们的实时、高分辨率HS-AFM成像捕获了ERα从单体到二聚体的结构转变,阐明了雌激素调节dna结合特异性的分子机制。基于这些发现,我们提出了一个配体诱导的二聚化(LID)模型,其中雌激素促进了ERα在DNA上的最佳负载。这些见解加深了我们对癌症中激素信号传导的理解,并为未来针对激素相关恶性肿瘤的治疗策略的发展带来了希望。本文章由计算机程序翻译,如有差异,请以英文原文为准。
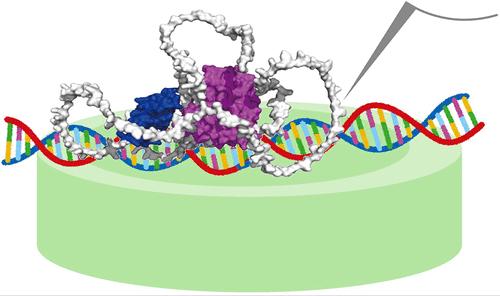
Zooming into Gene Activation: Estrogen Receptor α Dimerization and DNA Binding Visualized by High-Speed Atomic Force Microscopy
Estrogen receptor α (ERα) is pivotal in gene regulation, particularly in estrogen-responsive cancers. However, the full-length molecular dynamic structure of ERα remains elusive. In this study, we employ high-speed atomic force microscopy (HS-AFM) to visualize ERα interactions with the estrogen response element (ERE) under both ligand-present and ligand-absent conditions. ERα binds to ERE even in the absence of estrogen, although the presence of the ligand significantly enhances binding precision and stability. Our real-time, high-resolution HS-AFM imaging captures ERα structural transitions from monomeric to dimeric forms, elucidating the molecular mechanisms by which estrogen modulates DNA-binding specificity. Based on these findings, we propose a ligand-induced dimerization (LID) model, wherein estrogen facilitates the optimal loading of ERα onto DNA. These insights deepen our understanding of hormone signaling in cancer and hold promise for the development of future therapeutic strategies targeting hormone-related malignancies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Nano
工程技术-材料科学:综合
CiteScore
26.00
自引率
4.10%
发文量
1627
审稿时长
1.7 months
期刊介绍:
ACS Nano, published monthly, serves as an international forum for comprehensive articles on nanoscience and nanotechnology research at the intersections of chemistry, biology, materials science, physics, and engineering. The journal fosters communication among scientists in these communities, facilitating collaboration, new research opportunities, and advancements through discoveries. ACS Nano covers synthesis, assembly, characterization, theory, and simulation of nanostructures, nanobiotechnology, nanofabrication, methods and tools for nanoscience and nanotechnology, and self- and directed-assembly. Alongside original research articles, it offers thorough reviews, perspectives on cutting-edge research, and discussions envisioning the future of nanoscience and nanotechnology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


