氧化还原-中性光催化脱羧自由基加成/酰基迁移级联过程:模块化获得功能化α-(杂)芳基-α-氨基酮。
IF 2.7
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
本文以还原自由基-极性交叉为关键过程,成功地建立了一个稳健实用的N→C酰基迁移反应方案。在氧化还原-中性光催化作用下,多种酰胺类化合物可与羧酸反应生成α-(杂)芳基-α-氨基酮。这种脱羧自由基加成/酰基迁移级联过程具有底物范围广、功能相容性好、反应条件温和等特点。本文章由计算机程序翻译,如有差异,请以英文原文为准。
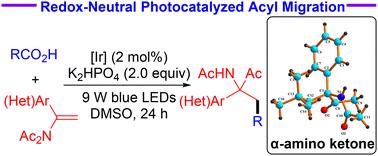
Redox-neutral-photocatalyzed decarboxylative radical addition/acyl migration cascade process: modular access to functionalized α-(hetero)aryl-α-aminoketones†
Herein, using reductive radical-polar crossover as a key process, a robust and practical protocol for the N → C acyl migration reaction has been successfully developed. A variety of enamides could react with carboxylic acids for modular access to α-(hetero)aryl-α-aminoketones enabled by redox-neutral photocatalysis. This decarboxylative radical addition/acyl migration cascade process features a broad substrate scope, good functional compatibility, and mild reaction conditions.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
Organic & Biomolecular Chemistry is an international journal using integrated research in chemistry-organic chemistry. Founded in 2003 by the Royal Society of Chemistry, the journal is published in Semimonthly issues and has been indexed by SCIE, a leading international database. The journal focuses on the key research and cutting-edge progress in the field of chemistry-organic chemistry, publishes and reports the research results in this field in a timely manner, and is committed to becoming a window and platform for rapid academic exchanges among peers in this field. The journal's impact factor in 2023 is 2.9, and its CiteScore is 5.5.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


