植物细胞周期调节器控制病毒致病的核环境
IF 18.7
1区 医学
Q1 MICROBIOLOGY
引用次数: 0
摘要
在病毒发病过程中,细胞周期调控因子的正确调控对病毒复制和寄主植物适应性生长都至关重要。reorchestrating RETINOBLASTOMA-RELATED 1 (RBR1), E2F转录因子的抑制因子和下游基因在宿主-病毒相互作用中的机制尚不清楚。在这里,我们发现番茄的后期促进复合物/环体(APC/C) E3连接酶激活细胞分裂周期20 (CDC20)结合RBR1或介导周期蛋白D1消耗以保存RBR1- e2f复合物,而双病毒或病毒重新利用APC/CCDC20活性以两种方式释放e2f:激活APC/CCDC20以消耗RBR1或阻断APC/CCDC20以刺激周期蛋白D1介导的RBR1消耗。释放的E2Fs激活DNA聚合酶或热休克蛋白70基因转录,有利于病毒繁殖。通过劫持APC/CCDC20不当破坏RBR1-E2F复合体导致宿主生长抑制。我们发现了一种情况,即病毒选择宿主APC/CCDC20重新编程RBR1-E2F复合体,以促进其繁殖,同时抑制宿主活力。本文章由计算机程序翻译,如有差异,请以英文原文为准。
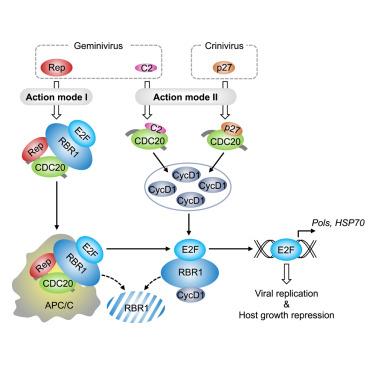
Plant cell-cycle regulators control the nuclear environment for viral pathogenesis
The proper regulation of cell-cycle regulators is curial for both viral replication and host-plant adaptive growth during the viral pathogenesis. Mechanisms on reorchestrating RETINOBLASTOMA-RELATED 1 (RBR1), repressor of E2F transcription factor, and downstream genes in host-virus interactions are unclear. Here, we discover that anaphase-promoting complex/cyclosome (APC/C) E3 ligase activator cell division cycle 20 (CDC20) in tomato binds RBR1 or mediates cyclin D1 depletion to preserve RBR1-E2F complexes, while geminivirus or crinivirus repurposes APC/CCDC20 activities to liberate E2Fs in two ways: activating APC/CCDC20 to deplete RBR1 or blocking APC/CCDC20 to stimulate cyclin-D1-mediated RBR1 depletion. The liberated E2Fs activate DNA polymerase or heat shock protein 70 gene transcription to favor virus propagation. The improper disruption of RBR1-E2F complexes via hijacking APC/CCDC20 causes the host growth repression. We uncover a scenario in which the virus co-opts host APC/CCDC20 to reprogram RBR1-E2F complex to favor its propagation while dampening host vitality.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell host & microbe
生物-微生物学
CiteScore
45.10
自引率
1.70%
发文量
201
审稿时长
4-8 weeks
期刊介绍:
Cell Host & Microbe is a scientific journal that was launched in March 2007. The journal aims to provide a platform for scientists to exchange ideas and concepts related to the study of microbes and their interaction with host organisms at a molecular, cellular, and immune level. It publishes novel findings on a wide range of microorganisms including bacteria, fungi, parasites, and viruses. The journal focuses on the interface between the microbe and its host, whether the host is a vertebrate, invertebrate, or plant, and whether the microbe is pathogenic, non-pathogenic, or commensal. The integrated study of microbes and their interactions with each other, their host, and the cellular environment they inhabit is a unifying theme of the journal. The published work in Cell Host & Microbe is expected to be of exceptional significance within its field and also of interest to researchers in other areas. In addition to primary research articles, the journal features expert analysis, commentary, and reviews on current topics of interest in the field.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


