过氧化氢-亚硫酸盐深度氧化法超快降解饮用水中微量污染物
IF 11.3
1区 环境科学与生态学
Q1 ENGINEERING, ENVIRONMENTAL
Journal of Hazardous Materials
Pub Date : 2025-06-15
Epub Date: 2025-02-28
DOI:10.1016/j.jhazmat.2025.137790
引用次数: 0
摘要
基于亚硫酸盐(S(IV))的高级氧化工艺(AOPs)最近因其低毒性和成本效益而受到关注,成为过硫酸盐基AOPs的可行替代品。过氧化氢(H2O2)被广泛认为是一种有效的环境友好型氧化剂。基于对H2O2对S(IV)的过量化学计量消耗的观察,提出了一种新的H2O2/S(IV) AOP。该系统产生硫酸盐自由基(SO4•-)、羟基自由基(•OH)、超氧阴离子自由基(O2•-)、单线态氧(1O2)等多种活性物质(RSs),实现饮用水中微量污染物的快速降解。当H2O2和S(IV)的剂量分别设置为0.1 mM和1.0 mM时,H2O2/S(IV)系统产生的SO4•-、•OH、O2•-和1O2的浓度约为10-12、10-12、10-13和10-13 m。即使在含有碳酸氢盐(HCO3-)、氯化物(Cl-)和腐植酸(HA)的复杂水基质中,pH范围为3.0至11.0,也会出现这种情况。建立了模拟RS生成的动力学模型,并预测了H2O2/S(IV)体系中15种微量污染物的伪一阶降解速率常数(k)。理论计算表明,高EHOMO和低ΔE(即ELUMO - EHOMO)的微污染物更容易被降解。与UV/H2O2、UV/S(IV)、Fe2+/H2O2和Fe3+/S(IV)体系相比,H2O2/S(IV)体系对微量污染物的降解速度更快,k值高1 ~ 2个数量级。此外,H2O2/S(IV)系统在控制后续氯化过程中消毒副产物的生成方面更为有效,凸显了H2O2/S(IV)系统在饮用水处理中的应用潜力。本文章由计算机程序翻译,如有差异,请以英文原文为准。
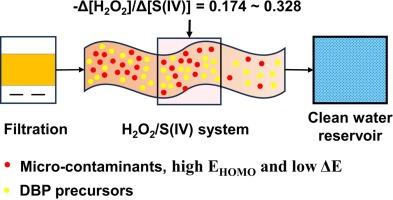
Advanced oxidation process with hydrogen peroxide and sulfite for superfast degradation of micro-contaminants in drinking water
Sulfite (S(IV))-based advanced oxidation processes (AOPs) have recently gained attention as viable alternatives to peroxosulfate-based AOPs due to their low toxicity and cost-effectiveness. Hydrogen peroxide (H2O2) is widely recognized as an effective and environmentally friendly oxidant in drinking water treatment. This study introduces a novel H2O2/S(IV) AOP based on the observation of over-stoichiometric consumption of S(IV) by H2O2. This system generates a variety of reactive species (RSs), including sulfate radicals (SO4•-), hydroxyl radicals (•OH), superoxide anion radicals (O2•–), and singlet oxygen (1O2), to achieve rapid degradation of micro-contaminants in drinking water. With dosages of H2O2 and S(IV) set at 0.1 mM and 1.0 mM, respectively, the H2O2/S(IV) system generated concentrations of SO4•-, •OH, O2•– and 1O2 at approximately 10−12, 10−12, 10−13, and 10−13 M. This occurred even in complex water matrices containing bicarbonate (HCO3-), chloride (Cl-), and humic acid (HA) across a pH range of 3.0–11.0. A kinetic model was developed to simulate RS generation and predict the pseudo-first-order degradation rate constants (k) for 15 micro-contaminants in the H2O2/S(IV) system. Theoretical calculations indicated that micro-contaminants with high EHOMO and low ΔE (i.e., ELUMO - EHOMO) are more susceptible to degradation. Compared to UV/H2O2, UV/S(IV), Fe2+/H2O2, and Fe3+/S(IV) systems, the H2O2/S(IV) system demonstrated faster degradation rates, with k values 1–2 orders of magnitude higher, towards micro-contaminants. Additionally, the H2O2/S(IV) system was more effective in controlling disinfection by-product formation during subsequent chlorination, highlighting the application potential of the H2O2/S(IV) system in drinking water treatment.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Hazardous Materials
工程技术-工程:环境
CiteScore
25.40
自引率
5.90%
发文量
3059
审稿时长
58 days
期刊介绍:
The Journal of Hazardous Materials serves as a global platform for promoting cutting-edge research in the field of Environmental Science and Engineering. Our publication features a wide range of articles, including full-length research papers, review articles, and perspectives, with the aim of enhancing our understanding of the dangers and risks associated with various materials concerning public health and the environment. It is important to note that the term "environmental contaminants" refers specifically to substances that pose hazardous effects through contamination, while excluding those that do not have such impacts on the environment or human health. Moreover, we emphasize the distinction between wastes and hazardous materials in order to provide further clarity on the scope of the journal. We have a keen interest in exploring specific compounds and microbial agents that have adverse effects on the environment.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



