用于同步干预糖酵解和氧化磷酸化以增强抗肿瘤疗法的氧调控酶纳米平台
IF 10.5
1区 医学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
本文章由计算机程序翻译,如有差异,请以英文原文为准。

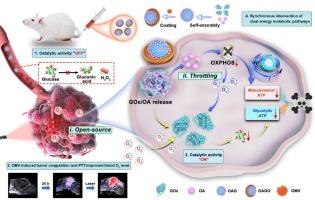
Oxygen-regulated enzymatic nanoplatform for synchronous intervention in glycolysis and oxidative phosphorylation to augment antitumor therapy
Tumor cells typically undergo metabolic reprogramming to obtain substantial energy via glycolysis and oxidative phosphorylation (OXPHOS). Intervening in this reprogramming is expected to have therapeutic effects, but simultaneous intervention in these two metabolic pathways is challenging. Herein, we developed an “open-source and throttling” oxygen (O₂) modulation strategy by which we can simultaneously intervene in these two metabolic pathways. Our O₂ modulation nanoplatform (denoted as OAGO) is fabricated via the self-assembly of glucose oxidase (GOx) and oligomycin A (OA) and is coated with bacterial outer membrane vesicles (OMVs). OAGO elicits simultaneous GOx-mediated inhibition of glycolysis and OA-induced inhibition of OXPHOS. The resulting production of GOx-catalyzed hydrogen peroxide leads to oxidative stress, which exacerbates the inhibition of mitochondrial function. Meanwhile, OA reduces intratumoral O₂ consumption (i.e., the “throttling” strategy), and OMVs increase the tumor blood O₂ level (i.e., the “open-source” strategy). This results in an increase in O₂ levels for GOx catalysis, thereby exacerbating energy consumption. In addition, OMVs increase intratumoral OAGO accumulation and enable photothermal therapy in the 4T1 mouse model, which also raises the tumor blood O₂ level and benefits GOx catalysis. This synchronous intervention in two metabolic pathways alongside O₂ modulation constitutes a promising approach for efficient antitumor therapy.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Controlled Release
医学-化学综合
CiteScore
18.50
自引率
5.60%
发文量
700
审稿时长
39 days
期刊介绍:
The Journal of Controlled Release (JCR) proudly serves as the Official Journal of the Controlled Release Society and the Japan Society of Drug Delivery System.
Dedicated to the broad field of delivery science and technology, JCR publishes high-quality research articles covering drug delivery systems and all facets of formulations. This includes the physicochemical and biological properties of drugs, design and characterization of dosage forms, release mechanisms, in vivo testing, and formulation research and development across pharmaceutical, diagnostic, agricultural, environmental, cosmetic, and food industries.
Priority is given to manuscripts that contribute to the fundamental understanding of principles or demonstrate the advantages of novel technologies in terms of safety and efficacy over current clinical standards. JCR strives to be a leading platform for advancements in delivery science and technology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


