IF 10.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
我们之前证明,长期训练免疫(TRIM)涉及在长寿命骨髓前体及其后代(单核细胞/巨噬细胞和中性粒细胞)中印刻先天性免疫记忆的适应性,从而增强对未来挑战的反应能力。在这里,我们展示了髓细胞生物学的一个独特组成部分--破骨细胞生成也可以接受先天性免疫训练。事实上,β-葡聚糖诱导的 TRIM 与骨髓中破骨细胞生成偏倚的增加和外周单核细胞/破骨细胞祖细胞的扩增有关,导致实验性牙周炎和关节炎的严重程度加剧。在训练有素的炎症性破骨细胞生成的背景下,我们观察到关节炎小鼠滑膜髓系细胞的转录组重构,其特点是转录因子黑色素生成相关转录因子(MITF)的显著上调。将经过β-葡聚糖训练的小鼠脾脏单核细胞收养性转移给天真受体,会以严格的MITF依赖性方式加重后者的关节炎。我们的研究结果表明,训练有素的破骨细胞生成是 TRIM 的不良适应成分,有可能为炎症性骨质流失疾病提供治疗靶点。本文章由计算机程序翻译,如有差异,请以英文原文为准。
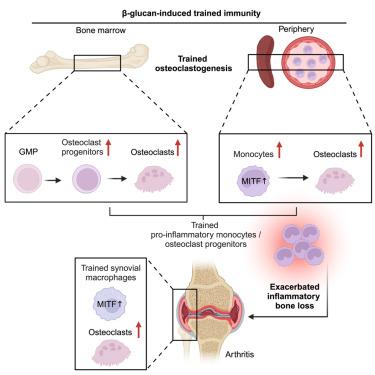
Innate immune training of osteoclastogenesis promotes inflammatory bone loss in mice
We previously demonstrated that long-term trained immunity (TRIM) involves adaptations that imprint innate immune memory in long-lived myelopoiesis precursors and their progeny, monocytes/macrophages and neutrophils, which thereby acquire enhanced responsiveness to future challenges. Here, we show that a distinct component of myeloid biology, osteoclastogenesis, can also undergo innate immune training. Indeed, β-glucan-induced TRIM was associated with an increased osteoclastogenesis bias in the bone marrow and an expansion of monocytes/osteoclast progenitors in the periphery, resulting in aggravated severity of experimental periodontitis and arthritis. In the setting of trained inflammatory osteoclastogenesis, we observed transcriptomic rewiring in synovial myeloid cells of arthritic mice, featuring prominent upregulation of the transcription factor melanogenesis-associated transcription factor (MITF). Adoptive transfer of splenic monocytes from β-glucan-trained mice to naive recipients exacerbated arthritis in the latter in a strictly MITF-dependent manner. Our findings establish trained osteoclastogenesis as a maladaptive component of TRIM and potentially provide therapeutic targets in inflammatory bone loss disorders.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Developmental cell
生物-发育生物学
CiteScore
18.90
自引率
1.70%
发文量
203
审稿时长
3-6 weeks
期刊介绍:
Developmental Cell, established in 2001, is a comprehensive journal that explores a wide range of topics in cell and developmental biology. Our publication encompasses work across various disciplines within biology, with a particular emphasis on investigating the intersections between cell biology, developmental biology, and other related fields. Our primary objective is to present research conducted through a cell biological perspective, addressing the essential mechanisms governing cell function, cellular interactions, and responses to the environment. Moreover, we focus on understanding the collective behavior of cells, culminating in the formation of tissues, organs, and whole organisms, while also investigating the consequences of any malfunctions in these intricate processes.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


