巨噬细胞保护周围神经病变的感觉轴突损失
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
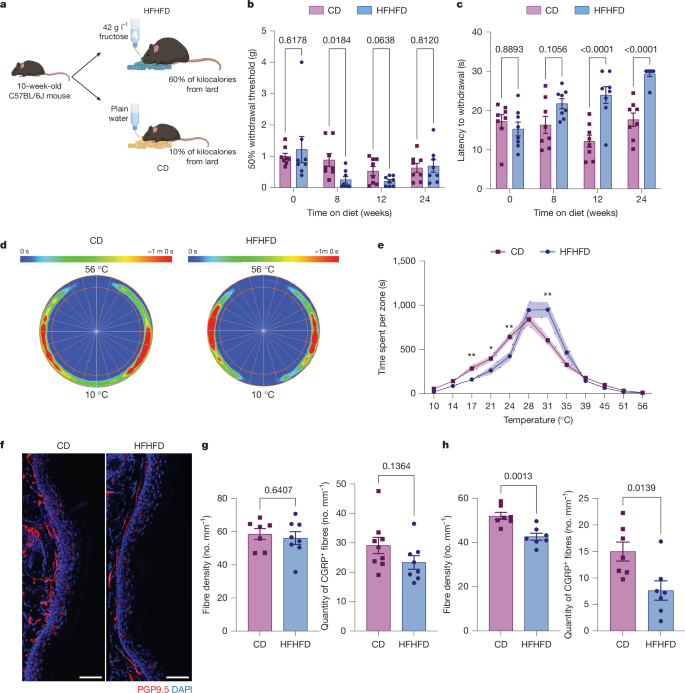
周围神经病变是2型糖尿病的常见并发症,它与肥胖密切相关1,引起感觉丧失,在一些患者中还会引起神经性疼痛2,3。虽然糖尿病周围神经病变的发生和发展与血脂异常和高血糖有关,但炎症对周围神经病变发病机制的贡献尚未得到研究。在这里,我们使用高脂肪,高果糖饮食(HFHFD),诱导肥胖和糖尿病前期代谢变化,研究周围神经病变的发病。饲喂HFHFD的小鼠在3个月后出现持续性热痛觉减退,但表皮皮肤神经纤维密度仅在6个月时出现减少。通过单细胞测序,我们发现CCR2+巨噬细胞在hhffd喂养小鼠的坐骨神经轴突变性检测到之前就已经浸润。这些浸润性巨噬细胞与神经挤压诱导的巨噬细胞具有相似的基因表达5,并表达神经变性相关的小胶质标记基因6,尽管没有轴突丢失或脱髓鞘。通过基因或药物阻断CCR2信号传导抑制巨噬细胞募集导致更严重的热痛觉减退和加速皮肤去神经,Lgals3基因的缺失也是如此,Lgals3基因在募集的巨噬细胞中表达。因此,巨噬细胞募集到肥胖糖尿病前期小鼠的周围神经具有神经保护作用,通过聚集素3延缓末梢感觉轴突变性。增强和维持患者早期的神经保护性免疫反应可以减缓或预防周围神经病变。本文章由计算机程序翻译,如有差异,请以英文原文为准。

Macrophages protect against sensory axon loss in peripheral neuropathy
Peripheral neuropathy is a common complication of type 2 diabetes, which is strongly associated with obesity1, causing sensory loss and, in some patients, neuropathic pain2,3. Although the onset and progression of diabetic peripheral neuropathy is linked with dyslipidaemia and hyperglycaemia4, the contribution of inflammation to peripheral neuropathy pathogenesis has not been investigated. Here we used a high-fat, high-fructose diet (HFHFD), which induces obesity and prediabetic metabolic changes, to study the onset of peripheral neuropathy. Mice fed the HFHFD developed persistent heat hypoalgesia after 3 months, but a reduction in epidermal skin nerve fibre density manifested only at 6 months. Using single-cell sequencing, we found that CCR2+ macrophages infiltrate the sciatic nerves of HFHFD-fed mice well before axonal degeneration is detectable. These infiltrating macrophages share gene expression similarities with nerve-crush-induced macrophages5 and express neurodegeneration-associated microglial marker genes6, although there is no axon loss or demyelination. Inhibiting the macrophage recruitment by genetically or pharmacologically blocking CCR2 signalling resulted in more severe heat hypoalgesia and accelerated skin denervation, as did deletion of Lgals3, a gene expressed in recruited macrophages. Recruitment of macrophages into the peripheral nerves of obese prediabetic mice is, therefore, neuroprotective, delaying terminal sensory axon degeneration by means of galectin 3. Potentiating and sustaining early neuroprotective immune responses in patients could slow or prevent peripheral neuropathy. A study in a mouse model of obesity and prediabetes demonstrates that recruitment of macrophages to nerves has a protective role in diet-induced peripheral neuropathy.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


