对中东呼吸综合征相关冠状病毒中ACE2使用情况的多个独立获取
IF 42.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
血管紧张素转换酶2 (ACE2)受体被具有不同受体结合域(RBD)结构的各种冠状病毒共享,但我们对这些趋同获取事件的理解仍然难以捉摸。在这里,我们报道了两种感染Pipistrellus nathusii (P.nat) -MOW15-22和pnnl2018b的蝙蝠mers相关冠状病毒(mersr - cov)使用ACE2作为其受体,具有狭窄的同源特异性。MOW15-22/PnNL2018B RBD-ACE2复合物的低温电子显微镜结构揭示了一种意想不到的、完全不同的结合模式,与任何其他已知的使用ace2的冠状病毒的结合模式>;45 Å不同。对105种哺乳动物的ACE2同源物进行功能分析,鉴定出了宿主趋向性决定因素,包括限制病毒识别的ACE2 n432糖基化,以及具有强病毒中和活性的可溶性P.nat ACE2突变体的设计。我们的研究结果揭示了在欧洲蝙蝠中发现的merbeco病毒的ACE2使用趋同性,强调了冠状病毒之间ACE2识别模式的非凡多样性和该受体的乱交性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
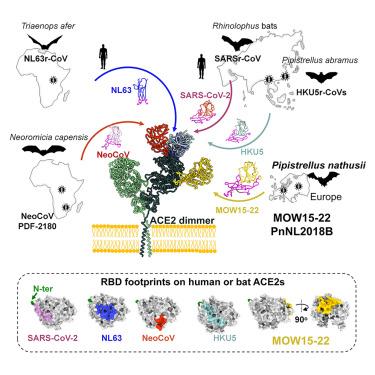
Multiple independent acquisitions of ACE2 usage in MERS-related coronaviruses
The angiotensin-converting enzyme 2 (ACE2) receptor is shared by various coronaviruses with distinct receptor-binding domain (RBD) architectures, yet our understanding of these convergent acquisition events remains elusive. Here, we report that two bat MERS-related coronaviruses (MERSr-CoVs) infecting Pipistrellus nathusii (P.nat)—MOW15-22 and PnNL2018B—use ACE2 as their receptor, with narrow ortholog specificity. Cryoelectron microscopy structures of the MOW15-22/PnNL2018B RBD-ACE2 complexes unveil an unexpected and entirely distinct binding mode, mapping >45 Å away from that of any other known ACE2-using coronaviruses. Functional profiling of ACE2 orthologs from 105 mammalian species led to the identification of host tropism determinants, including an ACE2 N432-glycosylation restricting viral recognition, and the design of a soluble P.nat ACE2 mutant with potent viral neutralizing activity. Our findings reveal convergent acquisition of ACE2 usage for merbecoviruses found in European bats, underscoring the extraordinary diversity of ACE2 recognition modes among coronaviruses and the promiscuity of this receptor.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


