Epstein-Barr病毒gp350介导补体受体结合和病毒中和的结构基础
IF 25.5
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
摘要
eb病毒(EBV)引起传染性单核细胞增多症,并与人类恶性肿瘤有关。B细胞的病毒感染是由病毒糖蛋白350 (gp350)与补体受体2 (CR2)结合引发的。尽管经过数十年的努力,仍未开发出疫苗或治疗药物,部分原因是缺乏对病毒-宿主界面的原子水平的了解。在这里,我们确定了gp350与CR2配合物的1.7 Å结构。gp350的CR2结合利用了识别其天然配体补体C3d所需的同一组Arg残基。我们进一步确定了gp350与三种有效中和抗体(nab)复合物的结构,这些抗体分别来自接种疫苗的猕猴和ebv感染者。与CR2相互作用一样,这些nab利用精氨酸残基靶向gp350上CR2结合位点内的酸性口袋。我们的研究结果阐明了分子模拟的两个轴——gp350对C3d和CR2对EBV抗体——为EBV疫苗和治疗方法的开发提供了见解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
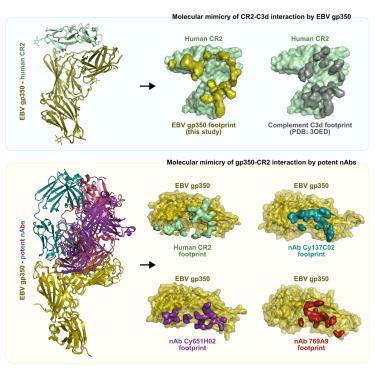
Structural basis for complement receptor engagement and virus neutralization through Epstein-Barr virus gp350
Epstein-Barr virus (EBV) causes infectious mononucleosis and is associated with malignancies in humans. Viral infection of B cells is initiated by the viral glycoprotein 350 (gp350) binding to complement receptor 2 (CR2). Despite decades of effort, no vaccines or curative agents have been developed, partly due to lack of atomic-level understanding of the virus-host interface. Here, we determined the 1.7 Å structure of gp350 in complex with CR2. CR2 binding of gp350 utilized the same set of Arg residues required for recognition of its natural ligand, complement C3d. We further determined the structures of gp350 in complex with three potently neutralizing antibodies (nAbs) obtained from vaccinated macaques and EBV-infected individuals. Like the CR2 interaction, these nAbs targeted the acidic pocket within the CR2-binding site on gp350 using Arg residues. Our results illustrate two axes of molecular mimicry—gp350 versus C3d and CR2 versus EBV nAbs—offering insights for EBV vaccines and therapeutics development.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Immunity
医学-免疫学
CiteScore
49.40
自引率
2.20%
发文量
205
审稿时长
6 months
期刊介绍:
Immunity is a publication that focuses on publishing significant advancements in research related to immunology. We encourage the submission of studies that offer groundbreaking immunological discoveries, whether at the molecular, cellular, or whole organism level. Topics of interest encompass a wide range, such as cancer, infectious diseases, neuroimmunology, autoimmune diseases, allergies, mucosal immunity, metabolic diseases, and homeostasis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


