母系X染色体影响雌性小鼠的认知和大脑老化
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
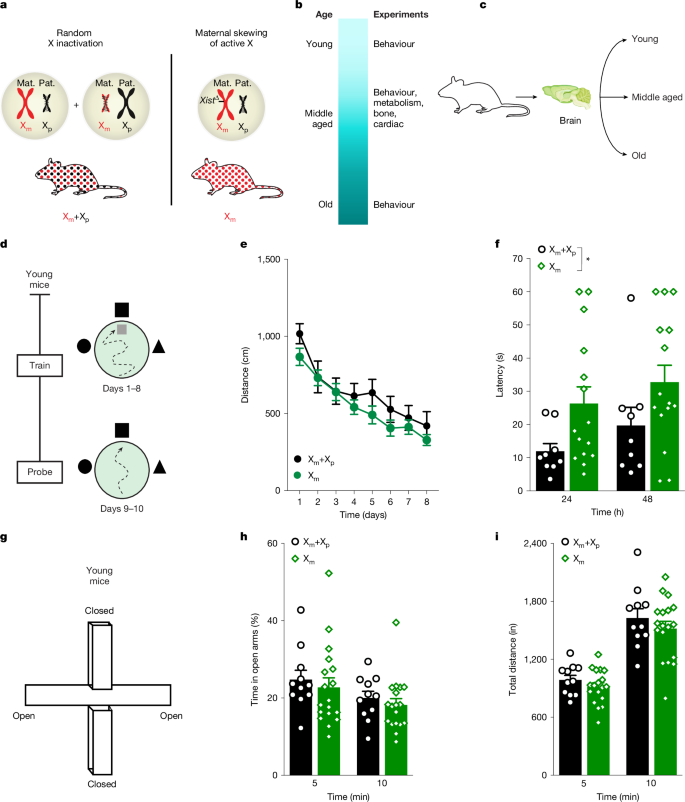
雌性哺乳动物细胞有两条X染色体,一条来自母系,一条来自父系。在发育过程中,一条X染色体随机失活(1,2,3,4)。这使得母体的X (Xm)染色体或父亲的X (Xp)染色体失活,导致雌性个体之间的X镶嵌现象不同,有些个体显示出相当大的或完全的X染色体歪斜,但仍然活跃5,6,7。双亲x源可以通过DNA甲基化8,9和可能的基因表达来改变表观遗传学;因此,嵌合可以缓冲衰老和疾病中的失调过程。然而,X偏或其镶嵌性是否会改变女性个体的功能在很大程度上是未知的。在这里,我们测试了向活跃的Xm染色体倾斜是否会影响大脑和身体,然后描绘了Xm神经元和Xp神经元的独特特征。活跃的Xm染色体损害了雌性小鼠整个生命周期的认知能力,并导致认知能力随着年龄的增长而恶化。在雌性小鼠中,认知缺陷伴随着xm介导的海马体生物或表观遗传老化加速,海马体是学习和记忆的关键中心。海马神经元的Xm染色体上有几个基因印迹,表明认知位点沉默。crispr介导的xm印迹基因激活改善了衰老雌性小鼠的认知能力。因此,Xm染色体损害了认知能力,加速了大脑衰老,并沉默了衰老过程中与认知有关的基因。了解Xm如何损害脑功能,可以更好地了解女性个体认知健康的异质性,以及防止认知缺陷和脑老化的x染色体衍生途径。本文章由计算机程序翻译,如有差异,请以英文原文为准。

The maternal X chromosome affects cognition and brain ageing in female mice
Female mammalian cells have two X chromosomes, one of maternal origin and one of paternal origin. During development, one X chromosome randomly becomes inactivated1–4. This renders either the maternal X (Xm) chromosome or the paternal X (Xp) chromosome inactive, causing X mosaicism that varies between female individuals, with some showing considerable or complete skew of the X chromosome that remains active5–7. Parent-of-X origin can modify epigenetics through DNA methylation8,9 and possibly gene expression; thus, mosaicism could buffer dysregulated processes in ageing and disease. However, whether X skew or its mosaicism alters functions in female individuals is largely unknown. Here we tested whether skew towards an active Xm chromosome influences the brain and body—and then delineated unique features of Xm neurons and Xp neurons. An active Xm chromosome impaired cognition in female mice throughout the lifespan and led to worsened cognition with age. Cognitive deficits were accompanied by Xm-mediated acceleration of biological or epigenetic ageing of the hippocampus, a key centre for learning and memory, in female mice. Several genes were imprinted on the Xm chromosome of hippocampal neurons, suggesting silenced cognitive loci. CRISPR-mediated activation of Xm-imprinted genes improved cognition in ageing female mice. Thus, the Xm chromosome impaired cognition, accelerated brain ageing and silenced genes that contribute to cognition in ageing. Understanding how Xm impairs brain function could lead to an improved understanding of heterogeneity in cognitive health in female individuals and to X-chromosome-derived pathways that protect against cognitive deficits and brain ageing. The maternal X chromosome in female mice impairs cognition through the silencing of certain genes and accelerates ageing of the hippocampus.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


