HMOX1-LDHB相互作用通过诱导晚期动脉粥样硬化中泡沫巨噬细胞线粒体功能障碍促进铁凋亡
IF 10.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
晚期动脉粥样硬化是急性心血管事件的病理基础,尽管采用现代治疗,仍有复发临床事件的显著残余风险。泡沫巨噬细胞的死亡是斑块进展的主要因素,但其潜在机制尚不清楚。大量和单细胞RNA测序表明,晚期动脉粥样硬化中大量铁积累促进泡沫巨噬细胞铁凋亡,特别是在髓样细胞2 (TREM2low)泡沫巨噬细胞中表达的触发受体低表达。该簇表现出低氧化磷酸化(OXPHOS)的代谢特征,增加了铁下垂的敏感性。机械上,血红素加氧酶1 (HMOX1)-乳酸脱氢酶B (LDHB)相互作用上调,使Lon肽酶1 (LONP1)降解线粒体转录因子A (TFAM),导致线粒体功能障碍和铁凋亡。给药线粒体靶向活性氧(ROS)清除剂MitoTEMPO(线粒体靶向TEMPO)或LONP1抑制剂硼替佐米恢复泡沫巨噬细胞线粒体稳态并减轻动脉粥样硬化。总的来说,我们的研究阐明了泡沫巨噬细胞铁质凋亡的细胞和分子机制,为晚期动脉粥样硬化提供了潜在的治疗策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
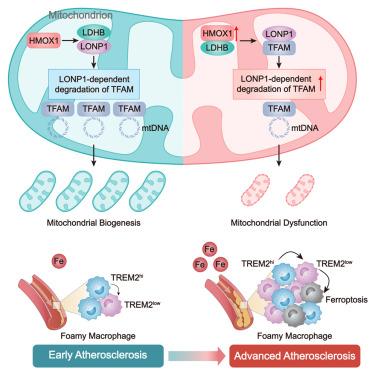
HMOX1-LDHB interaction promotes ferroptosis by inducing mitochondrial dysfunction in foamy macrophages during advanced atherosclerosis
Advanced atherosclerosis is the pathological basis for acute cardiovascular events, with significant residual risk of recurrent clinical events despite contemporary treatment. The death of foamy macrophages is a main contributor to plaque progression, but the underlying mechanisms remain unclear. Bulk and single-cell RNA sequencing demonstrated that massive iron accumulation in advanced atherosclerosis promoted foamy macrophage ferroptosis, particularly in low expression of triggering receptor expressed on myeloid cells 2 (TREM2low) foamy macrophages. This cluster exhibits metabolic characteristics with low oxidative phosphorylation (OXPHOS), increasing ferroptosis sensitivity. Mechanically, upregulated heme oxygenase 1 (HMOX1)-lactate dehydrogenase B (LDHB) interaction enables Lon peptidase 1 (LONP1) to degrade mitochondrial transcription factor A (TFAM), leading to mitochondrial dysfunction and ferroptosis. Administration of the mitochondria-targeted reactive oxygen species (ROS) scavenger MitoTEMPO (mitochondrial-targeted TEMPO) or LONP1 inhibitor bortezomib restored mitochondrial homeostasis in foamy macrophages and alleviated atherosclerosis. Collectively, our study elucidates the cellular and molecular mechanism of foamy macrophage ferroptosis, offering potential therapeutic strategies for advanced atherosclerosis.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Developmental cell
生物-发育生物学
CiteScore
18.90
自引率
1.70%
发文量
203
审稿时长
3-6 weeks
期刊介绍:
Developmental Cell, established in 2001, is a comprehensive journal that explores a wide range of topics in cell and developmental biology. Our publication encompasses work across various disciplines within biology, with a particular emphasis on investigating the intersections between cell biology, developmental biology, and other related fields. Our primary objective is to present research conducted through a cell biological perspective, addressing the essential mechanisms governing cell function, cellular interactions, and responses to the environment. Moreover, we focus on understanding the collective behavior of cells, culminating in the formation of tissues, organs, and whole organisms, while also investigating the consequences of any malfunctions in these intricate processes.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


