新系列3-吡啶-1,3-噻唑:体内和体外抗锥虫谱、体外和体内作用机制研究
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
锥虫病和利什曼病等锥虫科疾病是由锥虫科原生动物寄生虫引起的,它们分别是克鲁兹锥虫和利什曼原虫。目前迫切需要新的疗法。吡啶和噻唑环都是药物化学中公认的重要支架。本研究报告了 3-吡啶基-1,3-噻唑衍生物(1-18)的合成,并通过体外和体内试验对其进行了评估。对 T. cruzi、L. amazonensis 和 L. infantum 进行了体外试验,并使用 L929 成纤维细胞和 RAW 264.7 巨噬细胞评估了细胞毒性。作用模式研究包括体外试验和硅学模拟。14 个化合物具有杀锥虫活性,其 IC50 值在 0.2 至 3.9 μM 之间,优于苯并咪唑(4.2 μM)。化合物 7 的 IC50 值为 0.4 μM,选择性指数为 530.8。然而,当剂量为 100 毫克/千克/天时,这些化合物在体内试验中没有活性。化合物 1、7、8 和 10 具有抑制寄生虫、破坏线粒体、诱导细胞凋亡和破坏寄生虫膜的作用。这些化合物还能调节一氧化氮、IL-6、IL-10 和 TNF 的产生。硅学分析表明,这些化合物与克柔撒因有很强的相互作用,具有良好的生物利用度、药物相似性和稳定性。杀利什曼活性可忽略不计或不存在。尽管体外杀锥虫活性良好,但仍需要进一步的结构优化或制剂策略来提高口服稳定性和生物利用度,为开发新的治疗药物奠定基础。本文章由计算机程序翻译,如有差异,请以英文原文为准。
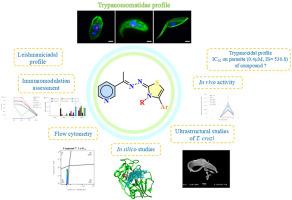

New series of 3-pyridyl-1,3-thiazoles: In vitro and in vivo anti-Trypanosomatidae profile, in vitro and in silico mechanism of action approach
Trypanosomatidae diseases, such as Chagas disease and leishmaniasis, are caused by protozoan parasites of the Trypanosomatidae family, namely Trypanosoma cruzi and Leishmania species, respectively. There is an urgent need for new therapies. Both pyridine and thiazole rings are recognized as important scaffolds in medicinal chemistry. This study reports the synthesis of 3-pyridyl-1,3-thiazole derivatives (1–18) and their evaluation through in vitro and in vivo assays. In vitro tests were conducted against T. cruzi, L. amazonensis, and L. infantum, with cytotoxicity assessed using L929 fibroblasts and RAW 264.7 macrophages. Mode of action studies included in vitro assays and in silico simulations. Fourteen compounds exhibited trypanocidal activity with IC50 values ranging from 0.2 to 3.9 μM, outperforming benznidazole (4.2 μM). Compound 7 displayed an IC50 of 0.4 μM and a selectivity index of 530.8. However, the compounds were inactive in vivo assays at a dose of 100 mg/kg/day. Compounds 1, 7, 8, and 10 demonstrated trypanostatic effects, mitochondrial disruption, apoptosis induction, and parasite membrane damage. These compounds also modulated nitric oxide, IL-6, IL-10 and TNF production. In silico analysis indicated strong interactions with cruzain and favorable bioavailability, drug-likeness, and stability profiles. The leishmanicidal activity was negligible or absent. Despite promising in vitro trypanocidal activity, further structural optimization or formulation strategies are required to enhance oral stability and bioavailability, providing a foundation for the development of new therapeutic agents.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


