早期肿瘤糖酵解下调预测三阴性乳腺癌患者对模拟禁食饮食的反应
IF 27.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
在临床前实验中,周期性禁食模拟饮食(FMDs)与化疗相结合显示出广泛的抗癌效果。在不同的肿瘤类型中,三阴性乳腺癌(TNBC)对 FMD 非常敏感。然而,循环 FMD 对 TNBC 患者的抗肿瘤活性和疗效仍不清楚。在此,我们展示了一种严重限制热量、每三周一次、为期 5 天的 FMD 方案,该方案与术前化疗相结合,可为 30 例参加 BREAKFAST 2 期试验的早期 TNBC 患者带来极佳的病理完全反应率(主要终点)和长期临床疗效(次要终点)。大量和单细胞RNA测序分析表明,获得pCR的肿瘤中的高糖酵解癌细胞、髓系细胞和周细胞在早期经历了与糖酵解和丙酮酸代谢相关的通路的显著下调。我们的研究结果为开展更大规模的临床试验铺平了道路,这些试验旨在研究周期性 FMD 对早期 TNBC 患者的疗效,并验证肿瘤内糖酵解的早期变化可作为营养限制临床获益的预测指标。该研究已在Clinicaltrials.gov(NCT04248998)上注册。本文章由计算机程序翻译,如有差异,请以英文原文为准。
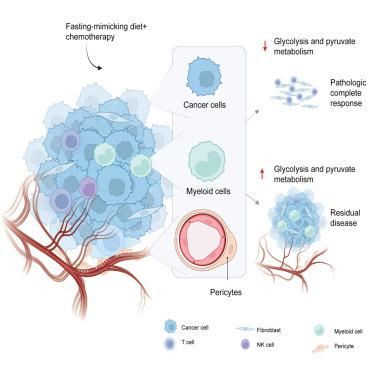
Early downmodulation of tumor glycolysis predicts response to fasting-mimicking diet in triple-negative breast cancer patients
In preclinical experiments, cyclic fasting-mimicking diets (FMDs) showed broad anticancer effects in combination with chemotherapy. Among different tumor types, triple-negative breast cancer (TNBC) is exquisitely sensitive to FMD. However, the antitumor activity and efficacy of cyclic FMD in TNBC patients remain unclear. Here, we show that a severely calorie-restricted, triweekly, 5-day FMD regimen results in excellent pathologic complete response (pCR) rates (primary endpoint) and long-term clinical outcomes (secondary endpoints) when combined with preoperative chemotherapy in 30 patients with early-stage TNBC enrolled in the phase 2 trial BREAKFAST. Bulk and single-cell RNA sequencing analysis revealed that highly glycolytic cancer cells, myeloid cells, and pericytes from tumors achieving pCR undergo a significant, early downmodulation of pathways related to glycolysis and pyruvate metabolism. Our findings pave the wave for conducting larger clinical trials to investigate the efficacy of cyclic FMD in early-stage TNBC patients and to validate early changes of intratumor glycolysis as a predictor of clinical benefit from nutrient restriction. This study was registered at Clinicaltrials.gov (NCT04248998).
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell metabolism
生物-内分泌学与代谢
CiteScore
48.60
自引率
1.40%
发文量
173
审稿时长
2.5 months
期刊介绍:
Cell Metabolism is a top research journal established in 2005 that focuses on publishing original and impactful papers in the field of metabolic research.It covers a wide range of topics including diabetes, obesity, cardiovascular biology, aging and stress responses, circadian biology, and many others.
Cell Metabolism aims to contribute to the advancement of metabolic research by providing a platform for the publication and dissemination of high-quality research and thought-provoking articles.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


