前列腺癌骨髓介导免疫治疗耐药的演变
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
晚期转移性去势抵抗性前列腺癌(mCRPC)患者对免疫检查点抑制剂(ICIs)难以耐受1,2,部分原因是肿瘤中存在免疫抑制髓细胞3,4。然而,骨髓细胞的异质性使得它们难以靶向,使得集落刺激因子-1受体(CSF1R)的阻断在临床上无效。在这里,我们对整个疾病连续体的患者活检使用单细胞谱分析,发现SPP1转录物水平升高的肿瘤相关巨噬细胞(spp1hi - tam)的独特群体随着前列腺癌向mCRPC的进展而变得丰富。在同基因小鼠模型中,类似的巨噬细胞群在体外抑制CD8+ T细胞活性,并在体内促进ICI抗性。此外,spp1hi - tam对抗csf1r抗体治疗无反应。途径分析确定腺苷信号是spp1hi - tam介导的免疫治疗耐药的潜在机制。事实上,在体外,腺苷A2A受体(A2ARs)的药理抑制显著逆转了sp1hi - tam介导的CD8+ T细胞免疫抑制,并在体内增强了CRPC对程序性细胞死亡蛋白1 (PD-1)阻断的反应性。与临床前结果一致,在mCRPC患者中,使用ciforadenant联合使用atezolizumab阻断程序性死亡1配体1 (PD-L1)来抑制A2ARs可诱导临床反应。此外,抑制A2ARs可导致CRPC中SPP1hi-TAM丰度显著降低,表明该途径既参与诱导,也参与下游免疫抑制。总之,这些发现表明spp1hi - tam是通过腺苷信号传导介导mCRPC中ICI耐药的关键介质,强调了spp1hi - tam作为治疗靶点和预测治疗效果的潜在生物标志物的重要性。本文章由计算机程序翻译,如有差异,请以英文原文为准。
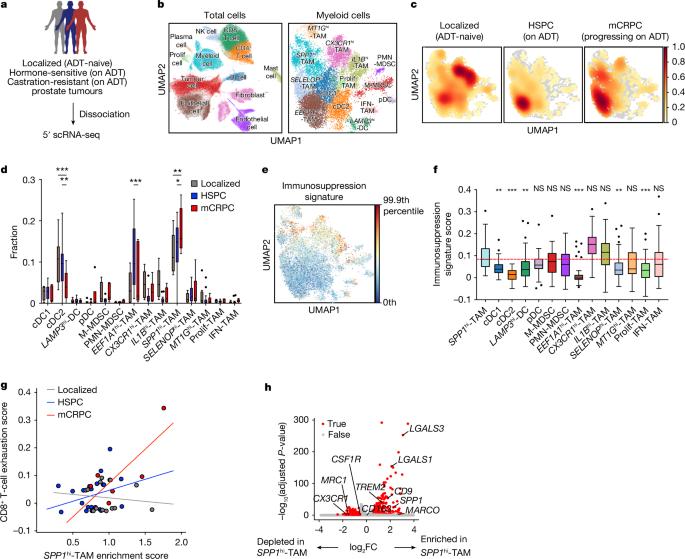
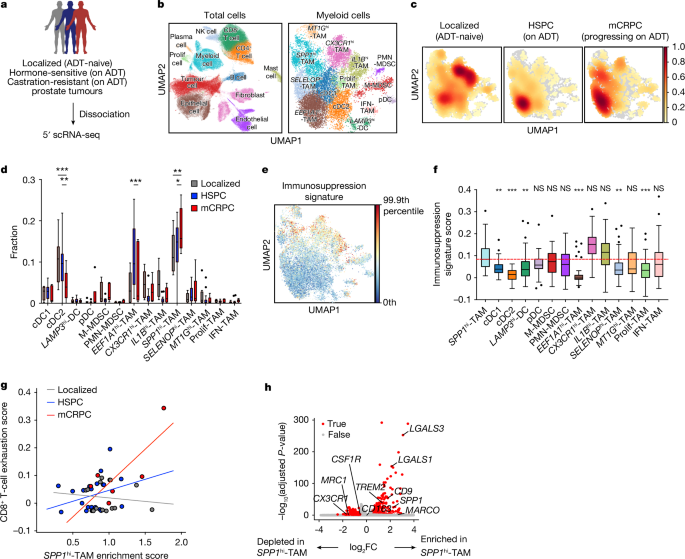
Evolution of myeloid-mediated immunotherapy resistance in prostate cancer
Patients with advanced metastatic castration-resistant prostate cancer (mCRPC) are refractory to immune checkpoint inhibitors (ICIs)1,2, partly because there are immunosuppressive myeloid cells in tumours3,4. However, the heterogeneity of myeloid cells has made them difficult to target, making blockade of the colony stimulating factor-1 receptor (CSF1R) clinically ineffective. Here we use single-cell profiling on patient biopsies across the disease continuum and find that a distinct population of tumour-associated macrophages with elevated levels of SPP1 transcripts (SPP1hi-TAMs) becomes enriched with the progression of prostate cancer to mCRPC. In syngeneic mouse modelling, an analogous macrophage population suppresses CD8+ T cell activity in vitro and promotes ICI resistance in vivo. Furthermore, Spp1hi-TAMs are not responsive to anti-CSF1R antibody treatment. Pathway analysis identifies adenosine signalling as a potential mechanism for SPP1hi-TAM-mediated immunotherapeutic resistance. Indeed, pharmacological inhibition of adenosine A2A receptors (A2ARs) significantly reverses Spp1hi-TAM-mediated immunosuppression in CD8+ T cells in vitro and enhances CRPC responsiveness to programmed cell death protein 1 (PD-1) blockade in vivo. Consistent with preclinical results, inhibition of A2ARs using ciforadenant in combination with programmed death 1 ligand 1 (PD-L1) blockade using atezolizumab induces clinical responses in patients with mCRPC. Moreover, inhibiting A2ARs results in a significant decrease in SPP1hi-TAM abundance in CRPC, indicating that this pathway is involved in both induction and downstream immunosuppression. Collectively, these findings establish SPP1hi-TAMs as key mediators of ICI resistance in mCRPC through adenosine signalling, emphasizing their importance as both a therapeutic target and a potential biomarker for predicting treatment efficacy. Single-cell profiling of human prostate cancer and studies in mouse models show that macrophages expressing SPP1 mediate immunotherapeutic resistance through adenosine pathway activation and represent a potential target for future studies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


