基于离子共价有机骨架的高性能锂金属电池准固态电解质
IF 4.1
2区 化学
Q2 POLYMER SCIENCE
引用次数: 0
摘要
锂金属固态电池由于其不可燃、耐高温、无腐蚀性等特性,在可充电储能设备中具有很大的应用前景。然而,它们的广泛应用受到锂离子电导率低和电极/电解质界面相容性差的阻碍。为了解决这些问题,我们合成了两个共价有机框架(COFs),一个是带有功能咪唑基团的(dha - cofilm),另一个是没有功能咪唑基团的(Dha-COF)。然后将离子液体(il)掺入这些COFs中,形成准固态电解质(dha - cofin - il和Dha-COF-IL)。Dha-COFim具有有序的多孔结构,形成相互连接的离子通道,可以快速传输锂离子并促进锂盐的解离,具有良好的热稳定性,高离子电导率(1.74 × 10⁻³cm⁻1),在室温下具有宽的电化学窗口。密度泛函理论(DFT)计算表明,dha - co薄膜中固定的咪唑基团增强了与TFSI毒血症的相互作用,促进了锂盐的解离。这使得自由锂离子能够以最小的能量损失快速通过通道。此外,在锂金属/电解质界面处形成了富含LiF和Li3N的稳定SEI层,加速了Li +的传输,确保了均匀的锂沉积和优异的电池性能。当与LiFePO4阴极结合时,LiFePO4‖Dha-COFim-IL‖锂电池提供高放电容量和出色的循环稳定性,为高能量密度锂电池设计准固态电解质提供了新的策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
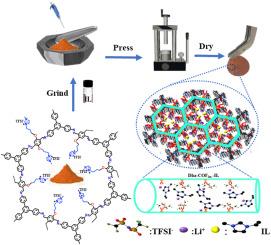

Ionic covalent organic framework based quasi-solid-state electrolyte for high-performance lithium metal battery
Lithium metal solid-state batteries are promising as rechargeable energy storage devices due to their non-combustible nature, resistance to high temperatures, and non-corrosive properties. However, their widespread application is hindered by low lithium-ion conductivity and poor compatibility at the electrode/electrolyte interface. To address these challenges, two covalent organic frameworks (COFs), one with functional imidazolium groups (Dha-COFim) and one without (Dha-COF), were synthesized. Ionic liquids (ILs) were then incorporated into these COFs to create quasi-solid-state electrolytes (Dha-COFim-IL and Dha–COF–IL). The Dha-COFim, with its ordered porous structure, forms interconnected ion channels that enable fast lithium-ion transport and enhance lithium salt dissociation, achieving excellent thermal stability, high ionic conductivity (1.74 × 10⁻³ S cm⁻1), and a wide electrochemical window at room temperature. Density functional theory (DFT) calculations showed that the fixed imidazolium groups in Dha-COFim enhance interactions with TFSI⁻ anions, improving lithium salt dissociation. This allows free lithium ions to move quickly through the channels with minimal energy loss. Additionally, the formation of a stable SEI layer rich in LiF and Li3N at the lithium metal/electrolyte interface accelerates Li⁺transport, ensuring uniform lithium deposition and superior battery performance. When combined with a LiFePO4 cathode, the LiFePO4‖Dha-COFim-IL‖Li cell delivers high discharge capacity and excellent cycling stability, providing a new strategy for designing quasi-solid-state electrolytes for high-energy-density lithium batteries.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Polymer
化学-高分子科学
CiteScore
7.90
自引率
8.70%
发文量
959
审稿时长
32 days
期刊介绍:
Polymer is an interdisciplinary journal dedicated to publishing innovative and significant advances in Polymer Physics, Chemistry and Technology. We welcome submissions on polymer hybrids, nanocomposites, characterisation and self-assembly. Polymer also publishes work on the technological application of polymers in energy and optoelectronics.
The main scope is covered but not limited to the following core areas:
Polymer Materials
Nanocomposites and hybrid nanomaterials
Polymer blends, films, fibres, networks and porous materials
Physical Characterization
Characterisation, modelling and simulation* of molecular and materials properties in bulk, solution, and thin films
Polymer Engineering
Advanced multiscale processing methods
Polymer Synthesis, Modification and Self-assembly
Including designer polymer architectures, mechanisms and kinetics, and supramolecular polymerization
Technological Applications
Polymers for energy generation and storage
Polymer membranes for separation technology
Polymers for opto- and microelectronics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


