通过铱催化的 N-H 插入级联合成功能化吲哚:萘胺与α-重氮羰基化合物的亲核环化反应
IF 3.6
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
研究人员开发了一种新型的铱催化萘胺和α-二氮羰基化合物的[3 + 2]环化反应,用于快速组装致密官能化的吲哚。这一新催化过程代表了通过胺的 N-H 插入进行分子内亲核环化级联的第一个实例。各种萘胺和α-二氮羰基化合物都能以极高的产率和极好的官能团耐受性获得。该反应生成了有价值的吲哚衍生物,从而能够快速获得其他方法难以获得的新型杂环类似物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
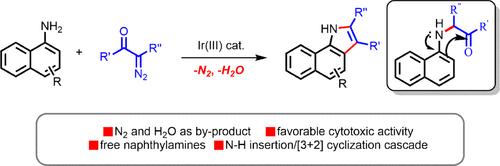
Synthesis of Functionalized Indoles by an Iridium-Catalyzed N–H Insertion Cascade: Nucleophilic Cyclization of Naphthylamines with α-Diazocarbonyl Compounds
A novel iridium-catalyzed [3 + 2] annulation of naphthylamines and α-diazocarbonyl compounds was developed for the rapid assembly of densely functionalized indoles. This new catalytic process represents the first example of a cascade intramolecular nucleophilic cyclization by the N–H insertion of amines. Various naphthylamines and α-diazocarbonyl compounds could be obtained in high yields with excellent functional group tolerance. The reaction affords valuable indole derivatives, enabling expedient access to novel heterocyclic analogues not easily accessible by other methods.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


