肠道生理学和环境可解释人类肠道微生物群组成和代谢的变化
IF 19.4
1区 生物学
Q1 MICROBIOLOGY
引用次数: 0
摘要
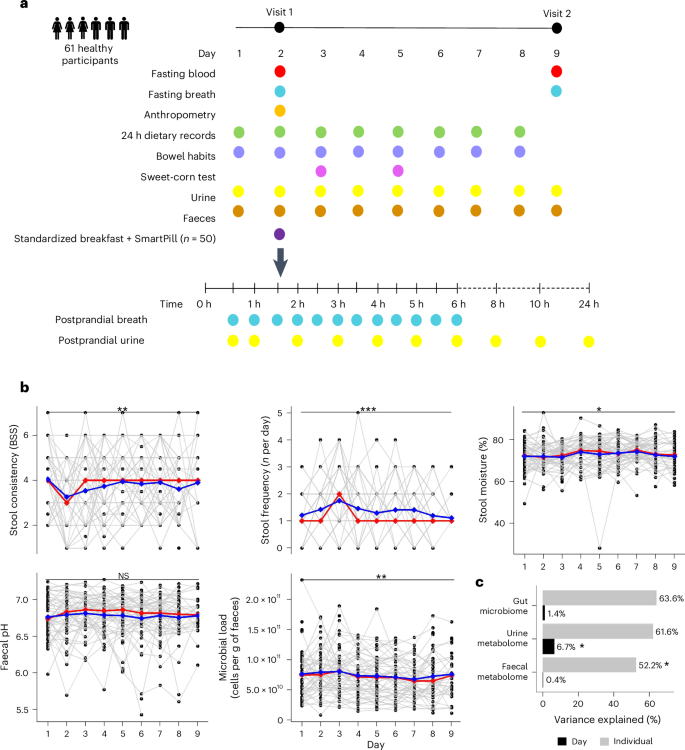
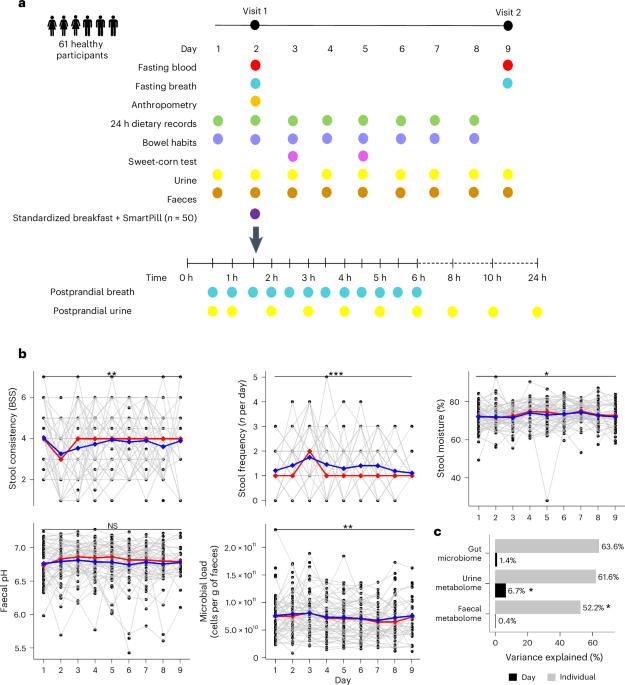
人类肠道微生物组是高度个性化的。然而,肠道生理和环境对肠道微生物组变化的影响仍未得到充分研究。在此,我们进行了一项观察性试验,利用多组学技术对 61 名健康成年人连续 9 天的微生物组组成和代谢情况进行了分析。我们评估了肠道环境因素的逐日变化,并使用无线蠕动胶囊测量了 50 人的全肠和节段肠道转运时间和 pH 值。我们观察到每天都有很大的波动,个体内肠道微生物群和新陈代谢的变化与粪便水分和粪便 pH 值的变化有关,而个体间的变化则由全肠和节段性肠道转运时间和 pH 值决定。微生物碳水化合物发酵产生的代谢物与肠道通过时间和 pH 值呈负相关,而蛋白水解代谢物和呼出甲烷呈正相关。最后,我们确定了节段通过时间/pH 与咖啡、饮食、宿主和微生物衍生代谢物之间的关联。我们的研究表明,肠道生理和环境是了解人类肠道微生物组成和新陈代谢个性的关键。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Gut physiology and environment explain variations in human gut microbiome composition and metabolism
The human gut microbiome is highly personal. However, the contribution of gut physiology and environment to variations in the gut microbiome remains understudied. Here we performed an observational trial using multi-omics to profile microbiome composition and metabolism in 61 healthy adults for 9 consecutive days. We assessed day-to-day changes in gut environmental factors and measured whole-gut and segmental intestinal transit time and pH using a wireless motility capsule in a subset of 50 individuals. We observed substantial daily fluctuations, with intra-individual variations in gut microbiome and metabolism associated with changes in stool moisture and faecal pH, and inter-individual variations accounted for by whole-gut and segmental transit times and pH. Metabolites derived from microbial carbohydrate fermentation correlated negatively with the gut passage time and pH, while proteolytic metabolites and breath methane showed a positive correlation. Finally, we identified associations between segmental transit time/pH and coffee-, diet-, host- and microbial-derived metabolites. Our work suggests that gut physiology and environment are key to understanding the individuality of the human gut microbial composition and metabolism. An observational longitudinal clinical trial, incorporating a SmartPill and metabolomics, reveals the role of host factors in shaping the gut microbiome in healthy human adults.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Microbiology
Immunology and Microbiology-Microbiology
CiteScore
44.40
自引率
1.10%
发文量
226
期刊介绍:
Nature Microbiology aims to cover a comprehensive range of topics related to microorganisms. This includes:
Evolution: The journal is interested in exploring the evolutionary aspects of microorganisms. This may include research on their genetic diversity, adaptation, and speciation over time.
Physiology and cell biology: Nature Microbiology seeks to understand the functions and characteristics of microorganisms at the cellular and physiological levels. This may involve studying their metabolism, growth patterns, and cellular processes.
Interactions: The journal focuses on the interactions microorganisms have with each other, as well as their interactions with hosts or the environment. This encompasses investigations into microbial communities, symbiotic relationships, and microbial responses to different environments.
Societal significance: Nature Microbiology recognizes the societal impact of microorganisms and welcomes studies that explore their practical applications. This may include research on microbial diseases, biotechnology, or environmental remediation.
In summary, Nature Microbiology is interested in research related to the evolution, physiology and cell biology of microorganisms, their interactions, and their societal relevance.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


