真菌内生菌产生的核苷信号可调控宿主细胞死亡并促进根系定殖
IF 20.6
1区 医学
Q1 MICROBIOLOGY
引用次数: 0
摘要
有益真菌内生菌 Serendipita indica 在植物根部的细胞内定植遵循一种双相策略,其中包括使拟南芥根部成功定植的宿主细胞死亡阶段。宿主细胞死亡是如何启动和控制的,目前尚不清楚。在这里,我们发现两种真菌酶,即外向-5′-核苷酸酶 SiE5NT 和核酸酶 SiNucA,在细胞死亡开始时协同作用于细胞外质,产生脱氧腺苷(dAdo)。细胞外 dAdo 而非结构相关的腺苷的摄取会通过平衡核苷转运体 ENT3 激活细胞死亡。我们发现了一种以前未定性的 Toll 样白细胞介素 1 受体(TIR)-核苷酸结合富亮氨酸重复受体(NLR)蛋白 ISI(由 S. indica 诱导),它是一种影响宿主细胞死亡、真菌定殖和生长促进的细胞内因子。我们的数据表明,两种真菌凋亡酶的联合活性促进了一种代谢物的产生,这种代谢物通过 TIR-NLR 调节的途径诱导植物细胞死亡,从而为植物的免疫代谢提供了一种联系。本文章由计算机程序翻译,如有差异,请以英文原文为准。
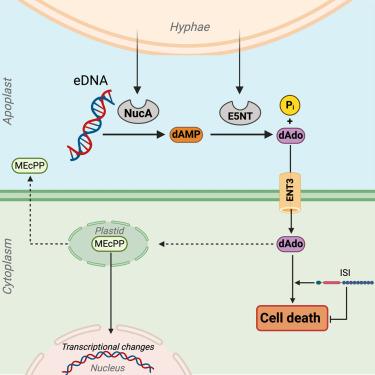
A nucleoside signal generated by a fungal endophyte regulates host cell death and promotes root colonization
The intracellular colonization of plant roots by the beneficial fungal endophyte Serendipita indica follows a biphasic strategy, including a host cell death phase that enables successful colonization of Arabidopsis thaliana roots. How host cell death is initiated and controlled is largely unknown. Here, we show that two fungal enzymes, the ecto-5′-nucleotidase SiE5NT and the nuclease SiNucA, act synergistically in the apoplast at the onset of cell death to produce deoxyadenosine (dAdo). The uptake of extracellular dAdo but not the structurally related adenosine activates cell death via the equilibrative nucleoside transporter ENT3. We identified a previously uncharacterized Toll-like interleukin 1 receptor (TIR)-nucleotide-binding leucine-rich repeat receptor (NLR) protein, ISI (induced by S. indica), as an intracellular factor that affects host cell death, fungal colonization, and growth promotion. Our data show that the combined activity of two fungal apoplastic enzymes promotes the production of a metabolite that engages TIR-NLR-modulated pathways to induce plant cell death, providing a link to immunometabolism in plants.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell host & microbe
生物-微生物学
CiteScore
45.10
自引率
1.70%
发文量
201
审稿时长
4-8 weeks
期刊介绍:
Cell Host & Microbe is a scientific journal that was launched in March 2007. The journal aims to provide a platform for scientists to exchange ideas and concepts related to the study of microbes and their interaction with host organisms at a molecular, cellular, and immune level. It publishes novel findings on a wide range of microorganisms including bacteria, fungi, parasites, and viruses. The journal focuses on the interface between the microbe and its host, whether the host is a vertebrate, invertebrate, or plant, and whether the microbe is pathogenic, non-pathogenic, or commensal. The integrated study of microbes and their interactions with each other, their host, and the cellular environment they inhabit is a unifying theme of the journal. The published work in Cell Host & Microbe is expected to be of exceptional significance within its field and also of interest to researchers in other areas. In addition to primary research articles, the journal features expert analysis, commentary, and reviews on current topics of interest in the field.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


