ZnO 和 TiO2-P25 纳米粒子促进布洛芬的光降解:全面的动力学、反应机制和热力学研究
IF 6.3
2区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
摘要
研究了用于布洛芬(IBU)光降解的商用 TiO2-P25 和 ZnO 纳米粒子(NPs)的光催化活性、反应动力学、模型和热力学。光降解实验在紫外线照射下的间歇反应器中进行。通过改变反应温度、催化剂体积密度和 IBU 溶液的初始浓度,进一步研究了 TiO2-P25 和 ZnO NPs 在不同操作条件下的光降解性能并建立了模型。通过估算的动力学参数和统计信息,对实验数据的描述性动力学模型进行了检验,结果表明 TiO2-P25 的反应速率为一阶,而 ZnO NPs 与 IBU 的反应速率为二阶。TiO2-P25 和 ZnO NPs 的光降解机理分别被确定为 Langmuir-Hinshelwood 和 Eley-Rideal。对热力学参数,特别是吉布斯自由能、焓和熵的变化进行了评估,结果表明这些 NPs 具有高效的光降解性能。本文章由计算机程序翻译,如有差异,请以英文原文为准。
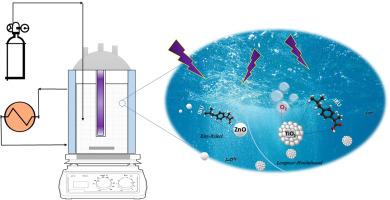
Ibuprofen photodegradation promoted by ZnO and TiO2-P25 nanoparticles: A comprehensive kinetic, reaction mechanisms, and thermodynamic investigation
Photocatalytic activity, reaction kinetics, modeling, and thermodynamics of commercial TiO2-P25 and ZnO nanoparticles (NPs) for ibuprofen (IBU) photodegradation were investigated. Photodegradation experiments were performed in a batch reactor under UV irradiation. The photodegradation performances of TiO2-P25 and ZnO NPs were further studied and modeled under different operation conditions, by varying the reaction temperature, catalyst bulk density, and the initial concentration of the IBU solution. The descriptive kinetic models for the experimental data were tested, through the estimated kinetic parameters, together with the statistical information, revealing that the reaction rate in the case of TiO2-P25 is of first order while the ZnO NPs follow second-order kinetics with respect to IBU. The photodegradation mechanisms for both TiO2-P25 and ZnO NPs were determined to be Langmuir-Hinshelwood and Eley-Rideal, respectively. Thermodynamic parameters were assessed, particularly, changes in Gibbs free energy, enthalpy, and entropy indicating the efficient photodegradation performance of these NPs.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of water process engineering
Biochemistry, Genetics and Molecular Biology-Biotechnology
CiteScore
10.70
自引率
8.60%
发文量
846
审稿时长
24 days
期刊介绍:
The Journal of Water Process Engineering aims to publish refereed, high-quality research papers with significant novelty and impact in all areas of the engineering of water and wastewater processing . Papers on advanced and novel treatment processes and technologies are particularly welcome. The Journal considers papers in areas such as nanotechnology and biotechnology applications in water, novel oxidation and separation processes, membrane processes (except those for desalination) , catalytic processes for the removal of water contaminants, sustainable processes, water reuse and recycling, water use and wastewater minimization, integrated/hybrid technology, process modeling of water treatment and novel treatment processes. Submissions on the subject of adsorbents, including standard measurements of adsorption kinetics and equilibrium will only be considered if there is a genuine case for novelty and contribution, for example highly novel, sustainable adsorbents and their use: papers on activated carbon-type materials derived from natural matter, or surfactant-modified clays and related minerals, would not fulfil this criterion. The Journal particularly welcomes contributions involving environmentally, economically and socially sustainable technology for water treatment, including those which are energy-efficient, with minimal or no chemical consumption, and capable of water recycling and reuse that minimizes the direct disposal of wastewater to the aquatic environment. Papers that describe novel ideas for solving issues related to water quality and availability are also welcome, as are those that show the transfer of techniques from other disciplines. The Journal will consider papers dealing with processes for various water matrices including drinking water (except desalination), domestic, urban and industrial wastewaters, in addition to their residues. It is expected that the journal will be of particular relevance to chemical and process engineers working in the field. The Journal welcomes Full Text papers, Short Communications, State-of-the-Art Reviews and Letters to Editors and Case Studies
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


