基于盐酸卡泊三醇酯的离子液体用于 GLP-1 胃给药:对体内性能的机理认识
IF 10.5
1区 医学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
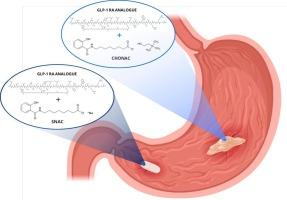
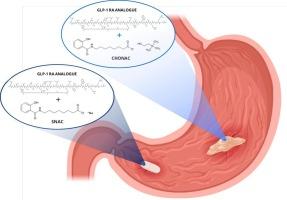
口服多肽需要使用高浓度渗透促进剂(PE)的配方来促进吸收,而且通常需要在服药和进食之间空腹一段时间。改进制剂以促进更快的吸收将增加使用的便利性,但需要更快的起效时间。我们开发了一种以盐己酸盐为基础的离子液体(IL)制剂,即盐己酸胆碱(CHONAC),用于通过胃吸收口服胰高血糖素样肽-1(GLP-1)类似物。体外研究证实,与参考片剂相比,在相同体积的剂型中可容纳更多的胰高血糖素,而且活性药物成分(API)和胰高血糖素的释放速度更快。实验证明,CHONAC制剂在4°C条件下的储存稳定性可达3周。首先在大鼠和麻醉犬体内对IL制剂的肽吸收功效进行了评估,结果表明与参考制剂相比,IL制剂的吸收速度更快。在清醒的狗体内,虽然CHONAC制剂仍能更早地吸收原料药,但其总体暴露量不如参考片剂。这主要归因于胃的生理机能,额外液体的存在会导致制剂稀释,以及液体快速进入十二指肠,在十二指肠中容易被蛋白水解的肽类药物(如本研究中使用的肽类药物)的吸收几乎可以忽略不计,这也可能是由于 CHONAC 在十二指肠区域的渗透增强能力较低。为了探讨这些问题,我们在麻醉狗身上进行了一项体内研究,研究涉及在胃中重复给药一种基于盐酸卡泊三醇酯的液体制剂,结果表明在整个给药期间都有可能以恒定的吸收率维持肽的吸收。总之,要想通过胃给药提高口服肽的生物利用度,就必须将 IL 制剂提供的高 PE 量和快速起效的优势结合起来,并确保肽和相关浓度的 PE 能与胃上皮细胞产生长时间的相互作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Salcaprozate-based ionic liquids for GLP-1 gastric delivery: A mechanistic understanding of in vivo performance
Oral delivery of peptides requires formulations with high concentrations of permeation enhancer (PE) to promote absorption, and often necessitates fasting time between dosing and food ingestion. Improved formulations promoting a more rapid absorption would increase convenience of use but requires a faster onset of action. We have developed a salcaprozate-based ionic liquid (IL) formulation, namely choline salcaprozate (CHONAC), for oral delivery of a glucagon-like peptide-1 (GLP-1) analogue via gastric absorption. In vitro studies confirmed the higher amount of PE accommodated in the same volume of dosage form as well as faster release of the active pharmaceutical ingredient (API) and PE compared to the tablet reference. Storage stability of the CHONAC formulation was demonstrated for up to 3 weeks at 4 °C. The peptide absorption efficacy of the IL formulation was first evaluated in vivo in rats and anesthetized dogs, showing a faster absorption compared to the reference formulations. In awake dogs, while the CHONAC formulation still enabled earlier API absorption, its overall exposure was inferior to the tablet reference. This was attributed mostly to the gastric physiology, causing formulation dilution in the presence of additional fluid as well as fast transit of liquids into the duodenum, where peptides liable to proteolytic degradation such as the one used in this study showed a negligible absorption, potentially also due to a lower permeation-enhancing capability of CHONAC in the duodenal region. Exploring these issues, an in vivo study in anesthetized dogs involving repeated dosing of a liquid salcaprozate-based formulation in the stomach revealed the potential to sustain peptide absorption throughout the dosing period with a constant absorption rate. In conclusion, combining the advantages of high PE amounts and fast onset of action provided by the IL formulation, and ensuring a prolonged interaction of peptide and PE at a relevant concentration with the stomach epithelium, are necessary to enhance oral peptide bioavailability via gastric delivery.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Controlled Release
医学-化学综合
CiteScore
18.50
自引率
5.60%
发文量
700
审稿时长
39 days
期刊介绍:
The Journal of Controlled Release (JCR) proudly serves as the Official Journal of the Controlled Release Society and the Japan Society of Drug Delivery System.
Dedicated to the broad field of delivery science and technology, JCR publishes high-quality research articles covering drug delivery systems and all facets of formulations. This includes the physicochemical and biological properties of drugs, design and characterization of dosage forms, release mechanisms, in vivo testing, and formulation research and development across pharmaceutical, diagnostic, agricultural, environmental, cosmetic, and food industries.
Priority is given to manuscripts that contribute to the fundamental understanding of principles or demonstrate the advantages of novel technologies in terms of safety and efficacy over current clinical standards. JCR strives to be a leading platform for advancements in delivery science and technology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


