通过 N-磺酰基-2-氨基苯甲醛和水杨醛衍生物与缺电子苄基氯的形式化 (4 + 1) 嵌合反应制备 C2-芳基吲哚和苯并呋喃。
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
两步形式(4 + 1)环化-脱水反应为获得 C2- 芳基化吲哚和苯并呋喃提供了一条便捷的途径。该反应利用了缺电子的苄基氯与 N-磺酰基-2-氨基苯甲醛或水杨醛衍生物的双功能反应性。该反应既可容忍取代醛上的吸电子基团和供电子基团,也可容忍苄基氯试剂对位吸电子基团的变化。这项研究还发现了这些苄基氯在碱性条件下自反应产生的有趣副产物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
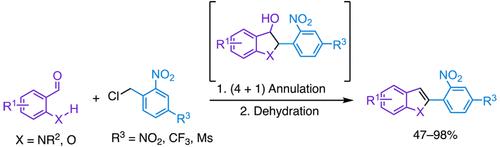
C2-Arylated Indoles and Benzofurans through Formal (4 + 1) Annulation of N-Sulfonyl-2-aminobenzaldehydes and Salicylaldehyde Derivatives with Electron-Deficient Benzyl Chlorides.
A two-step formal (4 + 1) annulation-dehydration reaction offers a convenient route to C2-arylated indoles and benzofurans. This reaction exploits the bifunctional reactivity of electron-deficient benzyl chlorides with N-sulfonyl-2-aminobenzaldehydes or salicylaldehyde derivatives. The reaction tolerates both electron-withdrawing and donating groups on the substituted aldehydes, as well as variation of electron-withdrawing groups at the para position of the benzyl chloride reagent. This work also identifies interesting byproducts resulting from the self-reaction of these benzyl chlorides under basic conditions.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


