发现一种用于治疗前列腺癌的强效 N 端域靶向 AR-FL/AR-V7 降解剂
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
以降解雄激素受体(AR)为靶点的 PROTACs 临床开发取得了重大进展。然而,对于含有雄激素受体剪接变体7(AR-V7)(一种没有配体结合域(LBD)的组成性活性突变体)的转移性前列腺癌,仍然缺乏有效的治疗方法。在此,我们报告了一种靶向 AR N-末端结构域(NTD)的高效力非共价 PROTAC NP18 的鉴定结果,NP18 由共价 AR-NTD 拮抗剂 EPI-002 发展而来,能有效降解 22Rv1 细胞中的 AR-FL 和 AR-V7(DC50 分别为 18 和 26 nM)。从机理上讲,NP18 与全长 AR(AR-FL)和剪接变体 7(AR-V7)的 N 端结构域(NTD)相互作用,导致它们被蛋白酶体选择性降解。重要的是,与 EPI-002 相比,NP18 在 22Rv1 异种移植和患者来源异种移植 (PDX) 模型中均表现出明显更强的抗肿瘤活性。综上所述,这些发现凸显了NP18是一种有希望对抗AR剪接变体驱动的耐药性的候选药物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
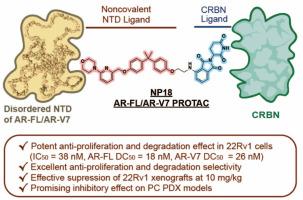
Discovery of a Highly Potent, N-terminal Domain-targeting degrader of AR-FL/AR-V7 for the treatment of Prostate Cancer
The clinical development of PROTACs targeting the androgen receptor (AR) for degradation has made significant progress. However, effective treatments for metastatic prostate cancers containing the androgen receptor splice variant 7 (AR-V7), a constitutively active mutant without the ligand-binding domain (LBD), are still lacking. Here, we reported the identification of a highly potent, noncovalent PROTAC targeting the N-terminal domain (NTD) of AR, NP18, which is developed from the covalent AR-NTD antagonist EPI-002, and effectively degrades both AR-FL and AR-V7 in 22Rv1 cells (DC50: 18 and 26 nM respectively). Mechanistically, NP18 interacts with the N-terminal domain (NTD) of both full-length AR (AR-FL) and splice variant 7 (AR-V7), leading to their selective and proteasomal degradation. Importantly, NP18 exhibited remarkably superior antitumor activity in both 22Rv1 xenograft and patient-derived xenograft (PDX) models than EPI-002. Taken together, these findings highlight NP18 as a promising candidate to counteract AR splice variant-driven resistance.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


