动态胺分选实现了不对称手性二胺的多选择性构建
IF 19.2
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
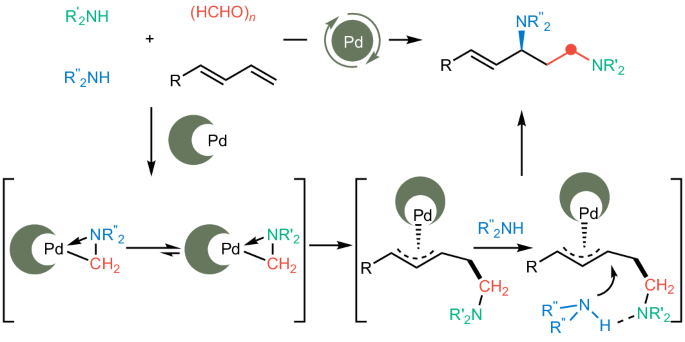
精确区分具有微小差异的化学物质是在化学合成和生物活性中实现高选择性的先决条件。然而,目前还缺乏对此类同族化合物进行区分和分类的有效策略,这给旨在有序加入这些化合物的合成工作带来了严峻的挑战。在此,我们报告了一种动态胺分类策略,该策略通过化学选择性形成胺甲基环钯化复合物来实现胺同系物的有效区分。一系列具有相似三维结构和性质的胺,以及众所周知的与金属的强结合能力,可以被有效地分化,从而实现二烯的高度化学、区域和对映体选择性多组分氨甲基化,以构建各种非对称手性二胺。这种动态胺分选策略解决了长期存在的难题,即如何精确区分和有序掺入具有微妙差异的脂肪族胺。从更广泛的角度来看,这一成功表明,精心设计的金属配合物可以为控制复杂合成中微妙的选择性提供灵活而通用的解决方案。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Dynamic amine sorting enables multiselective construction of unsymmetrical chiral diamines
Precisely differentiating chemicals featuring minor discrepancies is the prerequisite for achieving high selectivities in both chemical synthesis and biological activities. However, efficient strategies to differentiate and sort such congeneric compounds are lacking, posing daunting challenges for synthetic endeavours aimed at their orderly incorporation. Here we report a dynamic amine-sorting strategy that incorporates the chemoselective formation of the aminomethyl cyclopalladated complex to achieve the efficient differentiation of amine congeners. A series of amines sharing similar three-dimensional structures and properties, as well as possessing notoriously strong binding ability to metals, can be efficiently differentiated, enabling the highly chemo-, regio- and enantioselective multicomponent aminomethylamination of dienes to construct a variety of unsymmetrical chiral diamines. This dynamic amine-sorting strategy tackles the long-standing challenge of precise differentiation and orderly incorporation of aliphatic amines with subtle differences. From a broader perspective, the success demonstrates that meticulously designed metal complexes can provide flexible and general solutions for controlling delicate selectivities in sophisticated synthesis. Efficient strategies to differentiate and sort congeneric compounds are lacking. Now a metal-mediated dynamic amine-sorting strategy has been developed to precisely differentiate and assemble different amine congeners in an orderly manner, facilitating highly chemo-, regio- and enantioselective aminomethylamination of dienes for the construction of unsymmetrical chiral diamines.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature chemistry
化学-化学综合
CiteScore
29.60
自引率
1.40%
发文量
226
审稿时长
1.7 months
期刊介绍:
Nature Chemistry is a monthly journal that publishes groundbreaking and significant research in all areas of chemistry. It covers traditional subjects such as analytical, inorganic, organic, and physical chemistry, as well as a wide range of other topics including catalysis, computational and theoretical chemistry, and environmental chemistry.
The journal also features interdisciplinary research at the interface of chemistry with biology, materials science, nanotechnology, and physics. Manuscripts detailing such multidisciplinary work are encouraged, as long as the central theme pertains to chemistry.
Aside from primary research, Nature Chemistry publishes review articles, news and views, research highlights from other journals, commentaries, book reviews, correspondence, and analysis of the broader chemical landscape. It also addresses crucial issues related to education, funding, policy, intellectual property, and the societal impact of chemistry.
Nature Chemistry is dedicated to ensuring the highest standards of original research through a fair and rigorous review process. It offers authors maximum visibility for their papers, access to a broad readership, exceptional copy editing and production standards, rapid publication, and independence from academic societies and other vested interests.
Overall, Nature Chemistry aims to be the authoritative voice of the global chemical community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


