结构相似的卟啉和卟嗪在绿光或红光照射下对非黑素瘤皮肤肿瘤细胞的作用不同
IF 4.1
3区 工程技术
Q2 CHEMISTRY, APPLIED
引用次数: 0
摘要
我们合成了一种卟啉(H2P)和一种卟嗪(H2Pz)以及它们各自的锌配合物(ZnP 和 ZnPz),这些配合物的取代基为 N-乙基吡啶-3-基,并比较了它们的光物理和光毒性特性。由于含有阳离子分子,这些物质在水中的溶解度很高,而且在这种溶剂中没有明显的聚集现象。光物理研究表明,H2P 和 H2Pz 在水中的荧光量子产率(ΦF)分别为 2.04 % 和 0.48 %,金属化后,金属卟啉(ZnP)的荧光量子产率降至 1.4 %,金属卟嗪(ZnPz)的荧光量子产率增至 6.1 %。关于这些物质在水中产生单线态氧的效率(ΦΔ),卟啉与锌配位后从 16.2% 大幅增加到 41%,而卟嗪在这种溶剂中几乎不产生单线态氧(<1%)。在对人类鳞状细胞癌细胞(A-431)和永生化人类角质细胞(HaCaT)进行测试时,使用绿光(520 ± 5 nm)和红光(625 ± 5 nm)照射时,所有化合物的毒性都比未照射的细胞要高,但使用绿光时卟啉的光毒性更高,而卟嗪类化合物在红光照射下的表现更好。此外,卟啉的光毒性指数高于卟嗪,对 A-431 癌细胞的选择性也高于 HaCaT 细胞。据我们所知,这是第一项比较具有相同取代基的卟啉和卟嗪的光物理反应和体外抗癌活性的研究。本文章由计算机程序翻译,如有差异,请以英文原文为准。
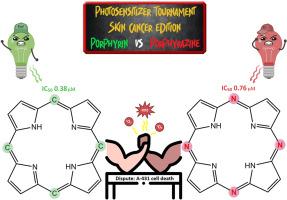
Structurally similar porphyrins and porphyrazines perform differently under green or red light irradiation against non-melanoma skin tumor cells
A porphyrin (H2P) and a porphyrazine (H2Pz) and their respective zinc complexes (ZnP and ZnPz), with the substituent N-ethylpyridyl-3-yl, were synthesized and their photophysical and phototoxic properties were compared. These substances showed high solubility in water due to the cationic moieties and without notable aggregation in this solvent. Photophysical studies showed that H2P and H2Pz have fluorescence quantum yields (ΦF) of 2.04 % and 0.48 % in water, respectively, that, upon metalation, decreases to 1.4 % for the metalloporphyrin (ZnP) and increases to 6.1 % for the metalloporphyrazine (ZnPz). Regarding the singlet oxygen production efficiency (ΦΔ) of the substances in water, a considerable increase from 16.2 % to 41 % was obtained for the porphyrin upon coordination to zinc, while the porphyrazines practically do not produce singlet oxygen (<1 %) in this solvent. When tested against human squamous cell carcinoma cells (A-431) and immortalized human keratinocytes (HaCaT), the toxicity of all compounds increased using green (520 ± 5 nm) and red (625 ± 5 nm) radiation when compared to the non-irradiated cells, however, phototoxicity was higher for the porphyrins using green light, while the porphyrazines performed better under red radiation. Furthermore, porphyrins had a higher phototoxic index than porphyrazines and greater selectivity for A-431 cancer cells compared to HaCaT cells. To the best of our knowledge, this is the first work that compares the photophysical response and in vitro anticancer activity of porphyrins and porphyrazines with the same substituent.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Dyes and Pigments
工程技术-材料科学:纺织
CiteScore
8.20
自引率
13.30%
发文量
933
审稿时长
33 days
期刊介绍:
Dyes and Pigments covers the scientific and technical aspects of the chemistry and physics of dyes, pigments and their intermediates. Emphasis is placed on the properties of the colouring matters themselves rather than on their applications or the system in which they may be applied.
Thus the journal accepts research and review papers on the synthesis of dyes, pigments and intermediates, their physical or chemical properties, e.g. spectroscopic, surface, solution or solid state characteristics, the physical aspects of their preparation, e.g. precipitation, nucleation and growth, crystal formation, liquid crystalline characteristics, their photochemical, ecological or biological properties and the relationship between colour and chemical constitution. However, papers are considered which deal with the more fundamental aspects of colourant application and of the interactions of colourants with substrates or media.
The journal will interest a wide variety of workers in a range of disciplines whose work involves dyes, pigments and their intermediates, and provides a platform for investigators with common interests but diverse fields of activity such as cosmetics, reprographics, dye and pigment synthesis, medical research, polymers, etc.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


