发现强效 PARP1 PROTAC 作为治疗结直肠癌的化疗增敏剂
IF 5.9
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
鉴于结直肠癌(CRC)患者很容易无法从化疗中持续获益,联合疗法经常被用作一种治疗策略。靶向 PARP1 阻断剂通过合成致死性对 BRCA1 或 BRCA2 突变的肿瘤细胞具有特异性毒性。本研究的重点是开发一系列靶向 PARP1 的强效 PROTACs,以提高具有 BRCA1 或 BRCA2 突变的 CRC 细胞对化疗的敏感性。通过对连接体进行精确的结构优化而得到的化合物 C6 能有效降解 PARP1,其 DC50 值为 58.14 nM。此外,C6 还大大提高了伊立替康的活性代谢物 SN-38 对 BRCA 基因突变的 CRC 细胞的细胞毒性,其有利的联合指数 (CI) 为 0.487。总之,这项研究强调了利用PAPRP1降解剂C6与伊立替康联合治疗携带BRCA突变的CRC患者的潜在益处。本文章由计算机程序翻译,如有差异,请以英文原文为准。
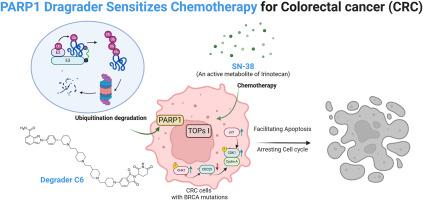
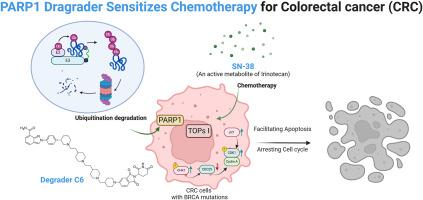
Discovery of a potent PARP1 PROTAC as a chemosensitizer for the treatment of colorectal cancer
Given the vulnerability of colorectal cancer (CRC) patients could not obtain a sustained benefit from chemotherapy, combination therapy is frequently employed as a treatment strategy. Targeting PARP1 blockade exhibit specific toxicity towards tumor cells with BRCA1 or BRCA2 mutations through synthetic lethality. This study focuses on developing a series of potent PROTACs targeting PARP1 in order to enhance the sensitivity of CRC cells with BRCA1 or BRCA2 mutations to chemotherapy. Compound C6, obtained based on precise structural optimization of the linker, has been shown to effectively degrade PARP1 with a DC50 value of 58.14 nM. Furthermore, C6 significantly increased the cytotoxic efficacy of SN-38, an active metabolite of Irinotecan, in BRCA-mutated CRC cells, achieving a favorable combination index (CI) of 0.487. In conclusion, this research underscores the potential benefits of employing a combination therapy that utilizes PAPRP1 degrader C6 alongside Irinotecan for CRC patients harboring BRCA mutations in CRC.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


