通过计算设计对单萜烯合成酶进行发散性改造以提高萜类化合物产量
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
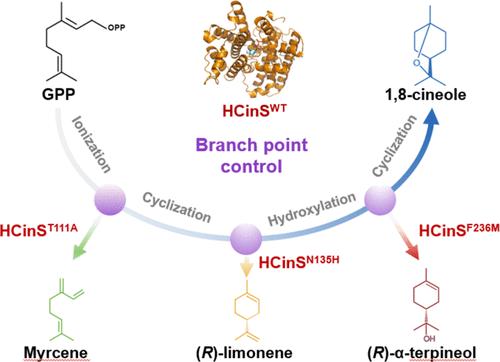
酶的催化杂合性使人们能够通过蛋白质工程改变产物的特异性;然而,利用这种杂合性来实现理想的催化反应仍然具有挑战性。在这里,我们发现了单萜烯合成酶(MTPS)HCinS,它对1,8-蒎烯的生物合成具有高效率和特异性。根据已解析的 HCinS 晶体结构进行的量子力学/分子力学(QM/MM)模拟揭示了生物合成级联反应的机理细节。在这些见解的指导下,我们设计了具有微调过渡态能量和反应微环境的硅学 HCinS 变体。三个变体(T111A、N135H 和 F236M)各有一个氨基酸取代,分别在生产单环 (R)-α-terpineol、(R)-limonene 和无环月桂烯时表现出高度特异性,保持了原生 HCinS 55% 以上的效率。这些设计的 HCinS 变体在催化能力上超过了自然进化的同工酶,并使酵母能够获得每种相应萜烯的最高微生物滴度。此外,在其他四种已确定的 TPS 中分别对四个功能相当的氨基酸进行单个突变,也会导致 HCinS 变体的产品特异性发生预期的转变。这项研究深入揭示了控制 TPS 产物杂交性的机制,并强调了计算设计在重塑 TPS 产物特异性方面的普遍适用性,从而为创造可应用于化学和合成生物学的酶铺平了创新之路。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Computational Design-Enabled Divergent Modification of Monoterpene Synthases for Terpenoid Hyperproduction
Enzymes’ catalytic promiscuity enables the alteration of product specificity via protein engineering; yet, harnessing this promiscuity to achieve desired catalytic reactions remains challenging. Here, we identified HCinS, a monoterpene synthase (MTPS) with a high efficiency and specificity for 1,8-cineole biosynthesis. Quantum mechanics/molecular mechanics (QM/MM) simulations, which were performed based on the resolved crystal structure of HCinS, revealed the mechanistic details of the biosynthetic cascade reactions. Guided by these insights, in silico HCinS variants were designed with fine-tuned transition-state energies and reaction microenvironments. Three variants (T111A, N135H, F236M), each with one amino acid substitution, exhibited high specificity in the production of monocyclic (R)-α-terpineol, (R)-limonene, and acyclic myrcene, respectively, maintaining over 55% efficiency of native HCinS. These designed HCinS variants surpassed naturally evolved isozymes in catalytic capacity and enabled yeast to achieve the highest microbial titer of each corresponding terpene. Furthermore, the single mutation of four functional equivalent amino acids in other four identified TPSs, respectively, resulted in the expected shifts on product specificity as HCinS variants. This research offers insights into the mechanisms controlling the TPS’s product promiscuity and highlights the universal applicability of computational design in reshaping the product specificity of TPSs, thereby paving innovative avenues for creating enzymes with applications in chemistry and synthetic biology.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


