来自完整噬菌体感染的 Synechococcus 的代谢物具有吸引海洋异养菌的化学作用
IF 20.5
1区 生物学
Q1 MICROBIOLOGY
引用次数: 0
摘要
化学线索介导了海洋浮游植物与细菌之间的相互作用,支撑着包括营养循环和碳固定在内的生态系统尺度过程。噬菌体感染会改变宿主的新陈代谢,刺激完整浮游生物释放化学线索,但人们对这些动态如何影响生态学和生物地球化学还知之甚少。在这里,我们利用时间分辨代谢组学和微流体技术确定了噬菌体感染对海洋蓝藻溶解代谢物池的影响以及异养细菌随后的趋化反应。从完整的、受噬菌体感染的Synechococcus中释放出的代谢物引起了藻溶弧菌和卤代假单胞菌的强烈趋化,尤其是在早期感染阶段。细菌对受活体感染的 Synechococcus 有持续的趋化作用,而对未感染的蓝藻则没有明显的趋化作用。高通量微流控技术确定了 5′-脱氧腺苷和 5′-甲硫腺苷为关键吸引物。我们的研究结果表明,受噬菌体感染的浮游微藻在溶解前会释放出能吸引异养细菌运动的化合物,这表明存在一种资源转移机制,可能会影响各营养级之间的碳和营养通量。本文章由计算机程序翻译,如有差异,请以英文原文为准。
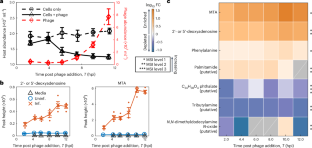

Metabolites from intact phage-infected Synechococcus chemotactically attract heterotrophic marine bacteria
Chemical cues mediate interactions between marine phytoplankton and bacteria, underpinning ecosystem-scale processes including nutrient cycling and carbon fixation. Phage infection alters host metabolism, stimulating the release of chemical cues from intact plankton, but how these dynamics impact ecology and biogeochemistry is poorly understood. Here we determine the impact of phage infection on dissolved metabolite pools from marine cyanobacteria and the subsequent chemotactic response of heterotrophic bacteria using time-resolved metabolomics and microfluidics. Metabolites released from intact, phage-infected Synechococcus elicited strong chemoattraction from Vibrio alginolyticus and Pseudoalteromonas haloplanktis, especially during early infection stages. Sustained bacterial chemotaxis occurred towards live-infected Synechococcus, contrasted by no discernible chemotaxis towards uninfected cyanobacteria. High-throughput microfluidics identified 5′-deoxyadenosine and 5′-methylthioadenosine as key attractants. Our findings establish that, before lysis, phage-infected picophytoplankton release compounds that attract motile heterotrophic bacteria, suggesting a mechanism for resource transfer that might impact carbon and nutrient fluxes across trophic levels. The authors use time-resolved metabolomics and microfluidics to characterize enhanced heterotroph chemoattraction to metabolites released from cyanobacteria during early stages of phage infection.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Microbiology
Immunology and Microbiology-Microbiology
CiteScore
44.40
自引率
1.10%
发文量
226
期刊介绍:
Nature Microbiology aims to cover a comprehensive range of topics related to microorganisms. This includes:
Evolution: The journal is interested in exploring the evolutionary aspects of microorganisms. This may include research on their genetic diversity, adaptation, and speciation over time.
Physiology and cell biology: Nature Microbiology seeks to understand the functions and characteristics of microorganisms at the cellular and physiological levels. This may involve studying their metabolism, growth patterns, and cellular processes.
Interactions: The journal focuses on the interactions microorganisms have with each other, as well as their interactions with hosts or the environment. This encompasses investigations into microbial communities, symbiotic relationships, and microbial responses to different environments.
Societal significance: Nature Microbiology recognizes the societal impact of microorganisms and welcomes studies that explore their practical applications. This may include research on microbial diseases, biotechnology, or environmental remediation.
In summary, Nature Microbiology is interested in research related to the evolution, physiology and cell biology of microorganisms, their interactions, and their societal relevance.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


