具有抗甲氧西林金黄色葡萄球菌靶向苯酚连接基团的新型 Pleuromutilin 衍生物的设计、合成和生物学评价
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
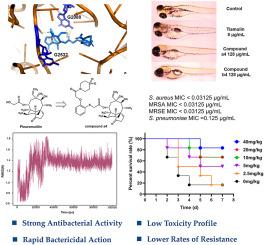
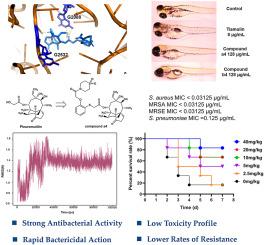
耐甲氧西林金黄色葡萄球菌(MRSA)仍然是全球健康的重大威胁,因此有必要开发新的治疗药物。Pleuromutilin 衍生物具有很强的抗菌活性,尤其是针对 MRSA 等革兰氏阳性细菌,因此是一种很有前景的解决方案。在这项研究中,我们合成了一系列带有含 C14 侧链的苯酚连接物的胸腺嘧啶衍生物,并对其体内外抗菌活性进行了评估。一些化合物对 MRSA 和金黄色葡萄球菌具有强效活性,最小抑菌浓度(MIC)低至 0.03125 μg/mL。特别是,与替姆林相比,化合物 a4 和 b4 显示出快速的杀菌活性,在时间杀伤动力学上显著减少了 MRSA 的载量,并显示出较慢的耐药性发展速度。在体内,与替姆林(ED50 = 11.38 mg/kg)相比,化合物 a4 在小鼠模型中以较低的有效剂量(ED50 = 6.48 mg/kg)减少 MRSA 引起的肺损伤方面表现出卓越的功效。分子对接和分子动力学研究还表明,化合物 a4 与核糖体肽基转移酶中心(PTC)结合力很强,而 PTC 是胸腺嘧啶衍生物的一个关键靶点。这些结果表明,化合物 a4 具有更强的抗菌活性和更低的耐药性,有望进一步开发成为治疗 MRSA 感染的有效药物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Design, synthesis, and biological evaluation of novel pleuromutilin derivatives with methicillin-resistant Staphylococcus aureus -targeting phenol linker groups
Methicillin-resistant Staphylococcus aureus (MRSA) remains a significant global health threat, necessitating the development of new therapeutic agents. Pleuromutilin derivatives offer a promising solution due to their potent antibacterial activity, particularly against Gram-positive bacteria such as MRSA. In this study, we synthesized a series of pleuromutilin derivatives with phenol linker containing C14 side chains and evaluated in vitro and in vivo antibacterial activities. Several compounds showed potent activity against MRSA and Staphylococcus aureus with minimal inhibitory concentrations (MICs) as low as 0.03125 μg/mL. In particular, compounds a4 and b4 showed rapid bactericidal activity, significantly reducing MRSA loads in time-kill kinetics and exhibiting slower resistance development compared to tiamulin. In vivo, compound a4 showed superior efficacy in reducing MRSA-induced lung damage in a mouse model at a lower effective dose (ED50 = 6.48 mg/kg) compared to tiamulin (ED50 = 11.38 mg/kg). Molecular docking and molecular dynamics studies also showed that compound a4 binds strongly to the ribosomal peptidyl transferase center (PTC), a key target for pleuromutilin derivatives. These results suggest that compound a4, with its enhanced antibacterial activity and low resistance potential, is a promising candidate for further development as an effective treatment for MRSA infections.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


