发现新型融合terocycle-bearing Diarypyrimidine Derivatives 作为 HIV-1 强效 NNRTIs,靶向耐受区 I 以增强抗病毒活性和耐药性特征
IF 5.9
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
作为抗艾滋病疗法的重要组成部分,HIV-1 非核苷类逆转录酶抑制剂饱受耐药性和毒性问题的困扰。我们以已报道的 XJ-18b1 为先导化合物,采用支架跳转策略设计了一系列新型二芳基嘧啶衍生物,发现了具有更强抗药性和类药物特征的强效 NNRTIs。活性最强的化合物 3k 对野生型 HIV-1(EC50 = 0.0019 μM)和常见突变株(包括 K103N(EC50 = 0.0019 μM)、L100I(EC50 = 0.0087 μM)、E138K(EC50 = 0.011 μM))具有显著的抑制活性,同时还具有低细胞毒性和高选择性指数(CC50 = 21.95 μM,SI = 11478)。此外,化合物 3k 对 HIV-2 具有抗病毒活性,EC50 值为 6.14 μM。酶联免疫吸附试验验证了 3k 能显著抑制 HIV-1 逆转录酶的活性(IC50 = 0.025 μM)。此外,还进行了分子动力学模拟研究,以说明 RT-3k 复合物的潜在结合模式和结合自由能。总之,3k 被证明是一种很有希望的先导化合物,可进一步优化以获得抗 HIV 候选药物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
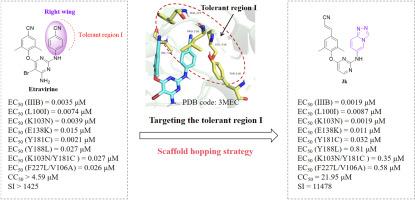
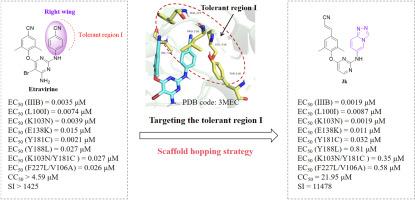
Discovery of novel fused-heterocycle-bearing diarypyrimidine derivatives as HIV-1 potent NNRTIs targeting tolerant region I for enhanced antiviral activity and resistance profile
As an important part of anti-AIDS therapy, HIV-1 non-nucleoside reverse transcriptase inhibitors are plagued by resistance and toxicity issues. Taking our reported XJ-18b1 as lead compound, we designed a series of novel diarypyrimidine derivatives by employing a scaffold hopping strategy to discover potent NNRTIs with improved anti-resistance properties and drug-like profiles. The most active compound 3k exhibited prominent inhibitory activity against wild-type HIV-1 (EC50 = 0.0019 μM) and common mutant strains including K103 N (EC50 = 0.0019 μM), L100I (EC50 = 0.0087 μM), E138K (EC50 = 0.011 μM), along with low cytotoxicity and high selectivity index (CC50 = 21.95 μM, SI = 11478). Additionally, compound 3k demonstrated antiviral activity against HIV-2 with EC50 value of 6.14 μM. The enzyme-linked immunosorbent assay validated that 3k could significantly inhibit the activity of HIV-1 reverse transcriptase (IC50 = 0.025 μM). Furthermore, molecular dynamics simulation studies were performed to illustrate the potential binding mode and binding free energy of the RT-3k complex, and in silico prediction revealed that 3k possessed favorable drug-like profiles. Collectively, 3k proved to be a promising lead compound for further optimization to obtain anti-HIV drug candidates.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


