用于无创诊断代谢功能障碍相关性脂肪性肝炎的血液生物标记物面板
IF 30.9
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
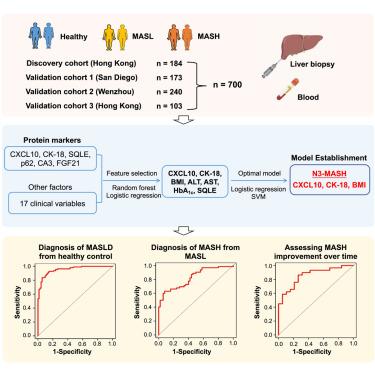
目前对代谢功能障碍相关性脂肪性肝病(MASLD)及其严重形式代谢功能障碍相关性脂肪性肝炎(MASH)的诊断并不理想。在这里,我们招募了 700 人,其中 184 人来自香港作为发现队列,516 人来自圣地亚哥、温州和香港作为三个验证队列。我们制定了一个由 3 个参数(C-X-C 矩阵趋化因子配体 10 [CXCL10]、细胞角蛋白 18 片段 M30 [CK-18]和调整后的体重指数 [BMI])组成的面板(称为 N3-MASH),该面板可将 MASLD 患者与健康对照组区分开来,接收者操作特征下面积 (AUROC) 为 0.954。在 MASLD 患者中,N3-MASH 可以识别 MASH 患者,其接收器操作特征下面积为 0.823,特异性为 90.0%,灵敏度为 62.9%,阳性预测值为 88.6%。N3-MASH 的诊断性能在三个验证队列中得到了证实,AUROC 分别为 0.802、0.805 和 0.823。此外,N3-MASH 还能识别 MASH 改善的患者,AUROC 为 0.857。总之,我们开发出了一种用于无创诊断 MASH 的稳健的血液面板,它可以帮助临床医生减少不必要的肝活检。本文章由计算机程序翻译,如有差异,请以英文原文为准。
A blood-based biomarker panel for non-invasive diagnosis of metabolic dysfunction-associated steatohepatitis
The current diagnosis of metabolic dysfunction-associated steatotic liver disease (MASLD) and its severe form, metabolic dysfunction-associated steatohepatitis (MASH), is suboptimal. Here, we recruited 700 individuals, including 184 from Hong Kong as a discovery cohort and 516 from San Diego, Wenzhou, and Hong Kong as three validation cohorts. A panel of 3 parameters (C-X-C motif chemokine ligand 10 [CXCL10], cytokeratin 18 fragments M30 [CK-18], and adjusted body mass index [BMI]) was formulated (termed N3-MASH), which discriminated patients with MASLD from healthy controls with an area under the receiver operating characteristic (AUROC) of 0.954. Among patients with MASLD, N3-MASH could identify patients with MASH with an AUROC of 0.823, achieving 90.0% specificity, 62.9% sensitivity, and 88.6% positive predictive value. The diagnostic performance of N3-MASH was confirmed in three validation cohorts with AUROC of 0.802, 0.805, and 0.823, respectively. Additionally, N3-MASH identifies patients with MASH improvement with an AUROC of 0.857. In summary, we developed a robust blood-based panel for the non-invasive diagnosis of MASH, which might help clinicians reduce unnecessary liver biopsies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell metabolism
生物-内分泌学与代谢
CiteScore
48.60
自引率
1.40%
发文量
173
审稿时长
2.5 months
期刊介绍:
Cell Metabolism is a top research journal established in 2005 that focuses on publishing original and impactful papers in the field of metabolic research.It covers a wide range of topics including diabetes, obesity, cardiovascular biology, aging and stress responses, circadian biology, and many others.
Cell Metabolism aims to contribute to the advancement of metabolic research by providing a platform for the publication and dissemination of high-quality research and thought-provoking articles.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


