III 型干扰素诱导肠道上皮细胞发生脓毒症并损害粘膜修复
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
组织损伤和修复是炎症的标志。尽管有大量关于组织损伤机制的信息,但对炎症如何影响修复的机制还缺乏深入了解。在这里,我们研究了干扰素如何影响肠粘膜损伤后的组织修复。我们发现,III 型而非 I 型或 II 型干扰素会通过诱导 Z-DNA 结合蛋白 1(ZBP1)的上调来延迟上皮细胞的再生。肠道损伤后形成的 Z 核酸会被 ZBP1 感知,从而导致 caspase-8 激活和 gasdermin C(GSDMC)的裂解。裂解后的 GSDMC 会导致上皮细胞热解死亡,并分别延缓结肠炎或辐照后大肠或小肠的修复。III 型干扰素/ZBP1/caspase-8/GSDMC 轴在炎症性肠病(IBD)患者中也很活跃。我们的研究结果突显了 III 型干扰素延迟肠道修复的能力,这对 IBD 患者或接受放射治疗的个体具有重要意义。本文章由计算机程序翻译,如有差异,请以英文原文为准。
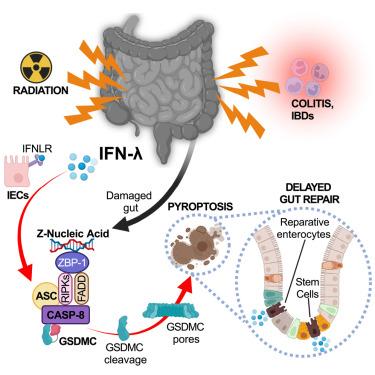
Type III interferons induce pyroptosis in gut epithelial cells and impair mucosal repair
Tissue damage and repair are hallmarks of inflammation. Despite a wealth of information on the mechanisms that govern tissue damage, mechanistic insight into how inflammation affects repair is lacking. Here, we investigated how interferons influence tissue repair after damage to the intestinal mucosa. We found that type III, not type I or type II, interferons delay epithelial cell regeneration by inducing the upregulation of Z-DNA-binding protein 1 (ZBP1). Z-nucleic acids formed following intestinal damage are sensed by ZBP1, leading to caspase-8 activation and the cleavage of gasdermin C (GSDMC). Cleaved GSDMC drives epithelial cell death by pyroptosis and delays repair of the large or small intestine after colitis or irradiation, respectively. The type III interferon/ZBP1/caspase-8/GSDMC axis is also active in patients with inflammatory bowel disease (IBD). Our findings highlight the capacity of type III interferons to delay gut repair, which has implications for IBD patients or individuals exposed to radiation therapies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


